Carboxypeptidase A
From Proteopedia
(Difference between revisions)
| Line 8: | Line 8: | ||
==Structure== | ==Structure== | ||
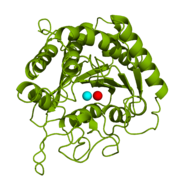
| - | Bovine CPA exists as a single unit with [http://en.wikipedia.org/wiki/Molecular_symmetry C1 symmetry] in the pancreatic physiological environment. According to [http://www.rcsb.org/pdb/explore/remediatedSequence.do?structureId=3CPA structural information] deposited in the PDB database, the single polypeptide chain of CPA contains a mixture of <scene name='69/694222/3cpasecondarystructure/2'>α-helices and β-sheets</scene>; there are a total of 11 helices: (one [http://en.wikipedia.org/wiki/310_helix 3<sub>10</sub>] and eight [http://en.wikipedia.org/wiki/Alpha_helix 3.6<sub>13</sub>]), as well as ten [http://en.wikipedia.org/wiki/Beta_sheet β-sheets]. The helices are shown in magenta whereas the β-sheets are displayed in yellow. A [http://en.wikibooks.org/wiki/Structural_Biochemistry/Chemical_Bonding/_Disulfide_bonds disulfide bond] connects the residues Cys138 and Cys161. The disulfide bond can be seen in yellow in the <scene name='69/694222/1cpx_default/3'>original rotating figure</scene>. | + | Bovine CPA exists as a single unit with [http://en.wikipedia.org/wiki/Molecular_symmetry C1 symmetry] in the pancreatic physiological environment. According to [http://www.rcsb.org/pdb/explore/remediatedSequence.do?structureId=3CPA structural information] deposited in the PDB database, the single polypeptide chain of CPA contains a mixture of <scene name='69/694222/3cpasecondarystructure/2'>α-helices and β-sheets</scene>; there are a total of 11 helices: (one [http://en.wikipedia.org/wiki/310_helix 3<sub>10</sub>] helix and eight [http://en.wikipedia.org/wiki/Alpha_helix 3.6<sub>13</sub>] helicies), as well as ten [http://en.wikipedia.org/wiki/Beta_sheet β-sheets]. The helices are shown in magenta whereas the β-sheets are displayed in yellow. A [http://en.wikibooks.org/wiki/Structural_Biochemistry/Chemical_Bonding/_Disulfide_bonds disulfide bond] connects the residues Cys138 and Cys161. The disulfide bond can be seen in yellow in the <scene name='69/694222/1cpx_default/3'>original rotating figure</scene>. |
| - | Six different biologically active forms of the CPA monomeric unit exist. <scene name='69/694222/1cpxcleavageforms/2'>Three of these active forms</scene> are produced following the cleavage of amino acid residue segments from the initial [http://en.wikipedia.org/wiki/Zymogen zymogen], or proenzyme, by trypsin and chymotrypsin, which are also found in the pancreas. Cleavage by trypsin generates either the '''α-form''' (residues Ala1-Asn307) or the '''β-form''' (residues Ser3-Asn307). Chymotrypsin cleavage generates the '''γ-form''' (residues Asn8-Asn307). The α-form essentially is the protein without any additional residue cleavages. The Ala and Arg residues, shown in red and white respectively, are cleaved in the β-form. In addition to the red and white residues, the residues displayed in yellow are cleaved to give the γ-form. The <scene name='69/694222/3cpageneticforms/3'>other three active forms</scene> of CPA arise from [http://en.wikipedia.org/wiki/Genetic_variation genetic variation] in residues located at three separate positions | + | Six different biologically active forms of the CPA monomeric unit exist. <scene name='69/694222/1cpxcleavageforms/2'>Three of these active forms</scene> are produced following the cleavage of amino acid residue segments from the initial [http://en.wikipedia.org/wiki/Zymogen zymogen], or proenzyme, by trypsin and chymotrypsin, which are also found in the pancreas. Cleavage by trypsin generates either the '''α-form''' (residues Ala1-Asn307) or the '''β-form''' (residues Ser3-Asn307). Chymotrypsin cleavage generates the '''γ-form''' (residues Asn8-Asn307). The α-form essentially is the protein without any additional residue cleavages. The Ala and Arg residues, shown in red and white respectively, are cleaved in the β-form. In addition to the red and white residues, the residues displayed in yellow are cleaved to give the γ-form. The <scene name='69/694222/3cpageneticforms/3'>other three active forms</scene> of CPA arise from [http://en.wikipedia.org/wiki/Genetic_variation genetic variation] in residues located at three separate positions on the polypeptide chain. The differences include the following: Ile/Val179, Ala/Glu228, and Val/Leu305.<ref name="CPA1" /> Each of the six biologically active monomeric units carry out the same function of hydrolyzing the C-terminal [http://en.wikipedia.org/wiki/Peptide_bond peptide bond] of a polypeptide substrate. |
===Active Site=== | ===Active Site=== | ||
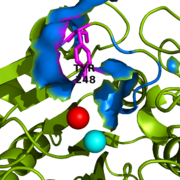
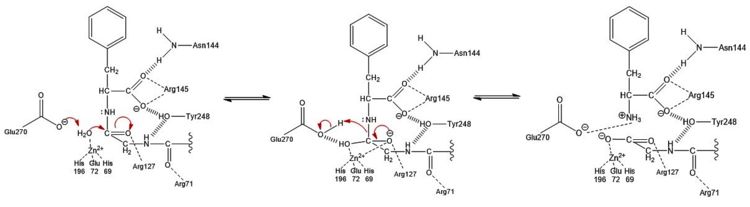
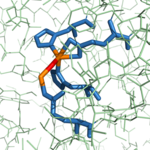
| - | The active site of bovine pancreatic CPA is embedded within a <scene name='69/694222/3cpadeeppocket/1'>deep pocket</scene> ( | + | The active site of bovine pancreatic CPA is embedded within a <scene name='69/694222/3cpadeeppocket/1'>deep pocket</scene> (shown in orange); the deep pocket's opening is located on the surface of the protein. Kinetics experiments have indicated that the binding region of the active site is actually capable of extending over five amino acids of the substrate.<ref name="CPA2" /> When no polypeptide substrate is bound in the active site, the pocket is open. However, the pocket is <scene name='69/694222/3cpadeeppocket2/2'>"capped" by a tyrosine residue (Tyr248)</scene> (see section titled "Important Tyr248 Residue") when a substrate or [http://en.wikipedia.org/wiki/Enzyme_inhibitor inhibitor] molecule binds.<ref name="CPA2" /> In this view, the Tyr248 is shown in green. The active site contains two separate subsites, labeled S1' and S1, each of which contain several pertinent residues that serve important roles during the catalyzed hydrolysis reaction. |
=====The Hydrophobic Binding Pocket: S1' Subsite===== | =====The Hydrophobic Binding Pocket: S1' Subsite===== | ||
Revision as of 02:12, 21 April 2017
| This Sandbox is Reserved from 02/09/2015, through 05/31/2016 for use in the course "CH462: Biochemistry 2" taught by Geoffrey C. Hoops at the Butler University. This reservation includes Sandbox Reserved 1051 through Sandbox Reserved 1080. |
To get started:
More help: Help:Editing |
Carboxypeptidase A in Bos taurus
| |||||||||||
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 Bukrinsky JT, Bjerrum MJ, Kadziola A. 1998. Native carboxypeptidase A in a new crystal environment reveals a different conformation of the important tyrosine 248. Biochemistry. 37(47):16555-16564. DOI: 10.1021/bi981678i
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 2.6 Christianson DW, Lipscomb WN. 1989. Carboxypeptidase A. Acc. Chem. Res. 22:62-69.
- ↑ Suh J, Cho W, Chung S. 1985. Carboxypeptidase A-catalyzed hydrolysis of α-(acylamino)cinnamoyl derivatives of L-β-phenyllactate and L-phenylalaninate: evidence for acyl-enzyme intermediates. J. Am. Chem. Soc. 107:4530-4535. DOI: 10.1021/ja00301a025
- ↑ Hirose, J., Noji, M., Kidani, Y., Wilkins, R. 1985. Interaction of zinc ions with arsanilazotyrosine-248 carboxypeptidase A.Biochemistry. 24(14):3495-3502. DOI:10.1021/bi00335a016
- ↑ Geoghegan, KF, Galdes, A, Martinelli, RA, Holmquist, B, Auld, DS, Vallee, BL. 1983. Cryospectroscopy of intermediates in the mechanism of carboxypeptidase A. Biochem. 22(9):2255-2262. DOI: 10.1021/bi00278a031
- ↑ Kaplan, AP, Bartlett, PA. 1991. Synthesis and evaluation of an inhibitor of carboxypeptidase A with a Ki value in the femtomolar range. Biochem. 30(33):8165-8170. PMID: 1868091
- ↑ Worthington, K., Worthington, V. 1993. Worthington Enzyme Manual: Enzymes and Related Biochemicals. Freehold (NJ): Worthington Biochemical Corporation; [2011; accessed March 28, 2017]. Carboxypeptidase A. http://www.worthington-biochem.com/COA/
- ↑ Pitout, MJ, Nel, W. 1969. The inhibitory effect of ochratoxin a on bovine carboxypeptidase a in vitro. Biochem. Pharma. 18(8):1837-1843. DOI: 0.1016/0006-2952(69)90279-2
- ↑ Normant, E, Martres, MP, Schwartz, JC, Gros, C. 1995. Purification, cDNA cloning, functional expression, and characterization of a 26-kDa endogenous mammalian carboxypeptidase inhibitor. Proc. Natl. Acad. Sci. 92(26):12225-12229. PMCID: PMC40329
Student Contributors
- Thomas Baldwin
- Michael Melbardis
- Clay Schnell
Proteopedia Page Contributors and Editors (what is this?)
Michael Melbardis, Douglas Schnell, Thomas Baldwin, Geoffrey C. Hoops, Michal Harel