User:Tilman Schirmer/Sandbox 211
From Proteopedia
| Line 36: | Line 36: | ||
<br><br> | <br><br> | ||
| - | + | ==Complete active site== | |
{{Theoretical_model}} | {{Theoretical_model}} | ||
After binding of the <scene name='User:Tilman_Schirmer/Sandbox_211/Competent_ggdef_dimer/6'>GTP substrate</scene> it is believed that two GGDEF domains associate antiparallely to form a catalytically competent dimer (<scene name='User:Tilman_Schirmer/Sandbox_211/Competent_ggdef_dimer/3'>view1</scene>, <scene name='User:Tilman_Schirmer/Sandbox_211/Competent_ggdef_dimer/1'>view2</scene>). Then, by inter-molecular nucleophilic in-line attack of the O3' atom onto the α-phosphorous of the other GTP substrate molecule two phosphodiester bonds are formed and 2 pyrophosphate molecules are released. | After binding of the <scene name='User:Tilman_Schirmer/Sandbox_211/Competent_ggdef_dimer/6'>GTP substrate</scene> it is believed that two GGDEF domains associate antiparallely to form a catalytically competent dimer (<scene name='User:Tilman_Schirmer/Sandbox_211/Competent_ggdef_dimer/3'>view1</scene>, <scene name='User:Tilman_Schirmer/Sandbox_211/Competent_ggdef_dimer/1'>view2</scene>). Then, by inter-molecular nucleophilic in-line attack of the O3' atom onto the α-phosphorous of the other GTP substrate molecule two phosphodiester bonds are formed and 2 pyrophosphate molecules are released. | ||
Revision as of 12:57, 21 June 2009
PleD
Overview
|
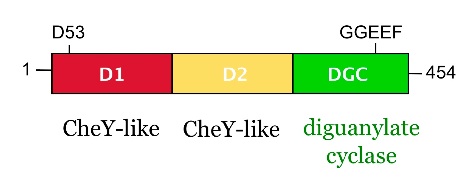
from Caulobacter crescentus is a response regulator with an unorthodox catalytic, diguanylate cyclase, output domain. It is composed of a canonical CheY-like response regulator receiver () domain,
a Rec-like () adaptor domain,
and a C-terminal domain that confers the catalytic acitvity.
The GGDEF domain is named after the highly conserved (in PleD it is GGEEF) that locates to a β-hairpin.
Substrate binding
|
The motif is part of the as identified in the structure of PleD in complex with . The GGDEFY domain binds only one GTP subsrate molecule. For the reaction to proceed, two GTP loaded GGDEF domains have to align antiparallely.
Complete active site
| Theoretical Model: The protein structure described on this page was determined theoretically, and hence should be interpreted with caution. |
After binding of the it is believed that two GGDEF domains associate antiparallely to form a catalytically competent dimer (, ). Then, by inter-molecular nucleophilic in-line attack of the O3' atom onto the α-phosphorous of the other GTP substrate molecule two phosphodiester bonds are formed and 2 pyrophosphate molecules are released.