User:Ramiro Barrantes/FpgNeiRepair
From Proteopedia
(Difference between revisions)
(→The FpgNei Protein Superfamily) |
(→The FpgNei Protein Superfamily) |
||
| Line 45: | Line 45: | ||
=== Functional Cluster 3: Stability of intercalation loop === | === Functional Cluster 3: Stability of intercalation loop === | ||
<p>[[Image:IntercalationLoopSupport.jpg|thumb|166px|right|This structure provides stability for the amino acids that insert into the space vacated by the damaged base]] The intercalation loop inserts into the DNA when the base is everted. This structure, as well as a group of amino acids that support it exhibit high conservation in Fpg, AY, plants and Neil1, but not on the rest. Note that the amino acid composition suggests that mimivirus has this structure.</p> | <p>[[Image:IntercalationLoopSupport.jpg|thumb|166px|right|This structure provides stability for the amino acids that insert into the space vacated by the damaged base]] The intercalation loop inserts into the DNA when the base is everted. This structure, as well as a group of amino acids that support it exhibit high conservation in Fpg, AY, plants and Neil1, but not on the rest. Note that the amino acid composition suggests that mimivirus has this structure.</p> | ||
| + | === Functional Cluster 5: Zinc/zincless finger === | ||
| + | <p>[[Image:ZincFingerjpg|thumb|166px|right|The Zinc Finger helps as a support of the key R264 residue]] The Zinc finger serves to support the Arg264 which binds to the phosphate of the damaged base. In Neil1, there is no Zinc but there is an equivalent structure <ref>PMID:15232006</ref>. Both the plants and mimivirus have a zincless finger, although it is not clear if this one is homologous</p> | ||
</div> | </div> | ||
<div style="float: left; width: 50%"> | <div style="float: left; width: 50%"> | ||
| Line 57: | Line 59: | ||
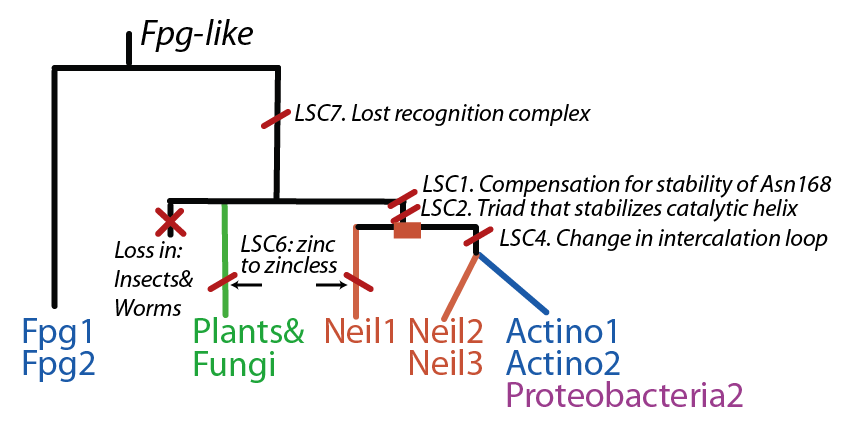
The FpgNei evolution has not been easy to resolve <ref>PMID:15232006</ref>, especially in the deeper branches. Assuming that functional clusters evolve more slowly than individual residues, we can use this as phylogenetic characters to 1) draw the most parsimonious evolution of the superfamily as dictated by these functional clusters 2) examine how these clusters have evolved and how this might have influenced the evolution of FpgNei. | The FpgNei evolution has not been easy to resolve <ref>PMID:15232006</ref>, especially in the deeper branches. Assuming that functional clusters evolve more slowly than individual residues, we can use this as phylogenetic characters to 1) draw the most parsimonious evolution of the superfamily as dictated by these functional clusters 2) examine how these clusters have evolved and how this might have influenced the evolution of FpgNei. | ||
[[Image:Scenario1.png]] | [[Image:Scenario1.png]] | ||
| - | |||
Revision as of 18:39, 21 May 2009
Contents |
The FpgNei Protein Superfamily
Functional Units
| G. Stereothermophilus Fpg | E. Coli Nei | ||||||||||||
|
|
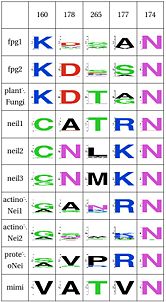
| Functional Cluster | Variant 1 | Variant 2 | Fpg1 | Fpg2 | Plant | Neil1 | Neil2 | Neil3 | Proteo | Actino1 | Actino2 | MimiVirus |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Support for perfectly conserved Asn168 | Y | Y | N | N | N | N | N | N | N | N | ||
| Stability of catalytic helix | Y | Y | Y | N | N | N | N | N | N | N | ||
| Stability of intercalation loop | Y | Y | Y | N | N | N | N | N | N | Y | ||
| Intercalation loop | Y | Y | Y | N | N | N | N | N | N | Y | ||
| Zinc finger | Y | Y | N | N | Y | Y | Y | Y | Y | N |
Functional Cluster 1: Stability of perfectly conserved Asn168
Asn74, along with two other amino acids have an effect in the orientation and kinking of the DNA. In 4 of the 9 clades (Fpg1, Fpg2 and Plants and Fungi) Asn174 is supported by Lys160, which in turn hydrogen bonds with Leu249 and Ser250. In the other clades (Actinobacteria 1 and 2, Proteacteria and all vertebrate sequences), Arg171 that comes from a different helix fulfills the same roles as Lys160. One important difference is that the Zinc Finger is shaped differently in the absence of DNA, and there is a hydrogen bond between one of the beta-sheets and the arginine. One hypothesis is that the arginine or the lysine is necessary to support the Asn174, crucial for orientation of the DNA.
Functional Cluster 3: Stability of intercalation loop
Image:IntercalationLoopSupport.jpg
This structure provides stability for the amino acids that insert into the space vacated by the damaged base
Functional Cluster 5: Zinc/zincless finger
Image:ZincFingerjpg
The Zinc Finger helps as a support of the key R264 residue
Functional Cluster 2: Stability of catalytic helix
The triad Leu4, Glu8 and Arg57 interact and provide stability to helixA, which has the catalytic residue Pro2,Glu3 and Glu6. This triad is present in the same four clades as above (Fpg1, Fpg2 and Plants and Fungi). This triad is not present in the remaining clades and it is not clear how the same stability is provided. Leu211 also has a hydrophobic interaction with Leu4.
Functional Cluster 4: Intercalation Loop
Image:IntercalationLoop.jpg
The residue in positions 77 and 78 suggest a possible intercalation loop
Evolution
The FpgNei evolution has not been easy to resolve [2], especially in the deeper branches. Assuming that functional clusters evolve more slowly than individual residues, we can use this as phylogenetic characters to 1) draw the most parsimonious evolution of the superfamily as dictated by these functional clusters 2) examine how these clusters have evolved and how this might have influenced the evolution of FpgNei.
References
- ↑ Doublie S, Bandaru V, Bond JP, Wallace SS. The crystal structure of human endonuclease VIII-like 1 (NEIL1) reveals a zincless finger motif required for glycosylase activity. Proc Natl Acad Sci U S A. 2004 Jul 13;101(28):10284-9. Epub 2004 Jul 1. PMID:15232006 doi:10.1073/pnas.0402051101
- ↑ Doublie S, Bandaru V, Bond JP, Wallace SS. The crystal structure of human endonuclease VIII-like 1 (NEIL1) reveals a zincless finger motif required for glycosylase activity. Proc Natl Acad Sci U S A. 2004 Jul 13;101(28):10284-9. Epub 2004 Jul 1. PMID:15232006 doi:10.1073/pnas.0402051101