Image:406322aa.2.jpg
From Proteopedia
No higher resolution available.
406322aa.2.jpg (600 × 415 pixel, file size: 47 KB, MIME type: image/jpeg)
Summary
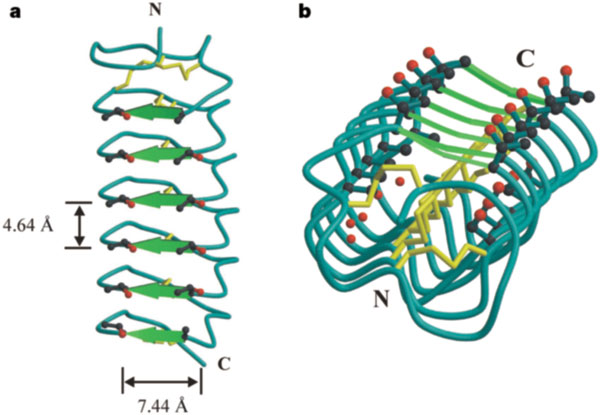
a: Side view of the TmAFP beta-helix with the beta-sheets (TCT sequences) indicated by green arrows and the disulphide bonds in yellow. Threonine side chains on the beta-sheet surface are shown with oxygen atoms in red. b: End-on view of the beta-helix with the N terminus proximal, showing the alignment of conserved threonine, cysteine, serine and alanine side chains and internal water. We note that the shorter dimension of the pseudo-rectangular cross-section in TmAFP is longer than the disulphide linkage, but the flatness of the beta-sheet is maintained by the opposite side being pulled inwards. The flatness of the beta-sheet is probably due to the shortness of the beta-strands, the disulphide bonds and the presence of favourable van der Waals interactions between stacked threonine side chains. The N and C termini are shown.
Licensing
{{subst:Permission from license selector}}
File history
Click on a date/time to view the file as it appeared at that time.
| Date/Time | User | Dimensions | File size | Comment | |
|---|---|---|---|---|---|
| (current) | 08:46, 19 January 2015 | Yulia Baron (Talk | contribs) | 600×415 | 47 KB | a, Side view of the TmAFP beta-helix with the beta-sheets (TCT sequences) indicated by green arrows and the disulphide bonds in yellow. Threonine side chains on the beta-sheet surface are shown with oxygen atoms in red. b, End-on view of the beta-helix wi |
| 08:18, 19 January 2015 | Yulia Baron (Talk | contribs) | 600×415 | 47 KB | a: Side view of the TmAFP beta-helix with the beta-sheets (TCT sequences) indicated by green arrows and the disulphide bonds in yellow. Threonine side chains on the beta-sheet surface are shown with oxygen atoms in red. b: End-on view of the beta-helix w |
- Edit this file using an external application
See the setup instructions for more information.
Links
The following pages link to this file:
