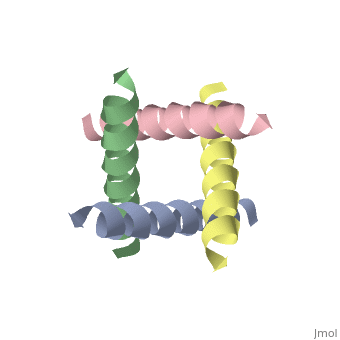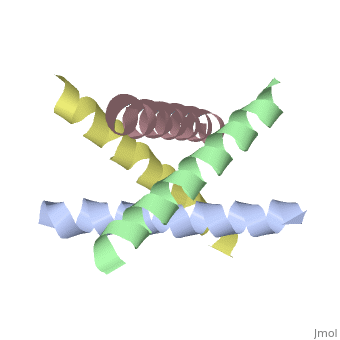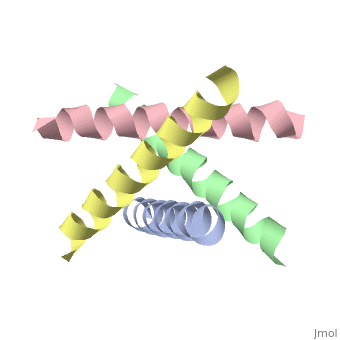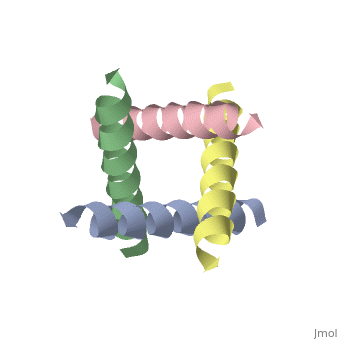This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
M2 Proton Channel
From Proteopedia
(→Background) |
(→Background) |
||
| Line 3: | Line 3: | ||
== Background == | == Background == | ||
| - | The M2 proton channel is a key protein that leads to viral infection [Takeuchi et al]. The M2 proton channel acidifies the viron which allows the viral matrix protein (M1) to disassociate from the ribonucleoprotein (RNP) [wu et al]. This allows the RNP to be transported to the nucleus of the cell [wu et al]. Several recent studies have looked at the effects of <scene name='User:Sarah_Henke/Sandbox_1/Amantadine/1'>amantadine</scene> and <scene name='User:Sarah_Henke/Sandbox_1/Rimantadine/1'>rimantadine</scene> on inhibiting the transfer of protons through the M2 channel [stouffer et al]. It has been found that M2 is resistant to these two drugs in 90% of humans, birds and pigs stouffer et al]. Understanding the structure and function of this proton channel is necessary in solving the resistance problem [stouffer et al]. | + | The M2 proton channel is a key protein that leads to viral infection [Takeuchi et al]. The M2 proton channel acidifies the viron which allows the viral matrix protein (M1) to disassociate from the ribonucleoprotein (RNP) [wu et al]. This allows the RNP to be transported to the nucleus of the cell [wu et al]. Several recent studies have looked at the effects of <scene name='User:Sarah_Henke/Sandbox_1/Amantadine/1'>amantadine</scene> [Stouffer et al] and <scene name='User:Sarah_Henke/Sandbox_1/Rimantadine/1'>rimantadine</scene> [Schnell et al] on inhibiting the transfer of protons through the M2 channel [stouffer et al]. It has been found that M2 is resistant to these two drugs in 90% of humans, birds and pigs stouffer et al]. Understanding the structure and function of this proton channel is necessary in solving the resistance problem [stouffer et al]. |
== Structure == | == Structure == | ||
Revision as of 21:37, 29 September 2009
Contents |
M2 Proton Channel from Influenza A Virus
|
Background
The M2 proton channel is a key protein that leads to viral infection [Takeuchi et al]. The M2 proton channel acidifies the viron which allows the viral matrix protein (M1) to disassociate from the ribonucleoprotein (RNP) [wu et al]. This allows the RNP to be transported to the nucleus of the cell [wu et al]. Several recent studies have looked at the effects of [Stouffer et al] and [Schnell et al] on inhibiting the transfer of protons through the M2 channel [stouffer et al]. It has been found that M2 is resistant to these two drugs in 90% of humans, birds and pigs stouffer et al]. Understanding the structure and function of this proton channel is necessary in solving the resistance problem [stouffer et al].
Structure
The M2 proton channel from influenza A is 97 amino acid residues and forms a 24-residue N-terminal extracellular domain, a 19-residue trans-membrane domain, and a 54-residue C-terminal cytoplasmic domain [wu et al]. The 19-residue TM domain forms the highly selective proton channel [Takashi et al]. Circular dichroism spectra has shown the TM domain to form one α-helix that spans the membrane [wu et al]. By analytical ultracentrifugation, the TM domain is found to form [takeuchi et al]. This tetrameric bundle of the TM domain is found by NMR to be tilted by 25-38° from the channel axis [takeuchi et al]. The TM helicies are arranged around the channel pore with an approximate fourfold rotational symmetry [takeuchi et al].
Central Cavity
|
pH Gating
References
Proteopedia Page Contributors and Editors (what is this?)
Sarah Henke, David Canner, Michal Harel, Alexander Berchansky, Eric Martz