This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox 156
From Proteopedia
(→Reaction of CAT III) |
(→Reaction of CAT III) |
||
| Line 14: | Line 14: | ||
[[Image:Picture2.jpg|500x400 px|center]] | [[Image:Picture2.jpg|500x400 px|center]] | ||
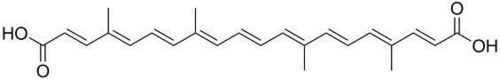
| - | In the first step of the reaction, Histidine-156 abstracts a proton from the 3-hydroxyl of chloramphenicol, promoting a nucleophilic attack from the deprotonated oxygen to the thioester bond of the acetyl-CoA. The intermediate produced, 3-acetylchloramphenicol, then rearranges non-enzymatically to 1-acetylchloramphenicol. Regeneration of the 3-hydroxyl allows another CAT III catalyzed nucleophilic attack to another acetyl-CoA and a 1,3-diacetylchloramphenicol product is formed. PMID:2015231 | + | In the first step of the reaction, Histidine-156 abstracts a proton from the 3-hydroxyl of chloramphenicol, promoting a nucleophilic attack from the deprotonated oxygen to the thioester bond of the acetyl-CoA. The intermediate produced, 3-acetylchloramphenicol, then rearranges non-enzymatically to 1-acetylchloramphenicol. Regeneration of the 3-hydroxyl allows another CAT III catalyzed nucleophilic attack to another acetyl-CoA and a 1,3-diacetylchloramphenicol product is formed. <ref>PMID:2015231</ref> |
==Structure== | ==Structure== | ||
Revision as of 04:56, 21 March 2010
Contents |
Chloramphenicol Acetyltransferase Type III
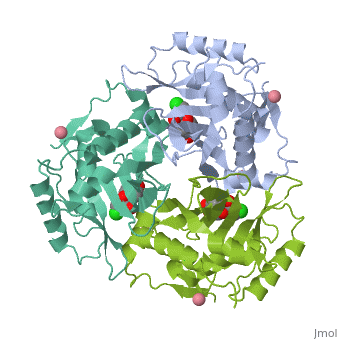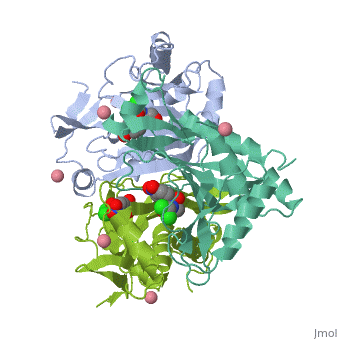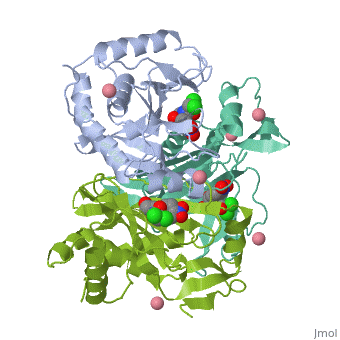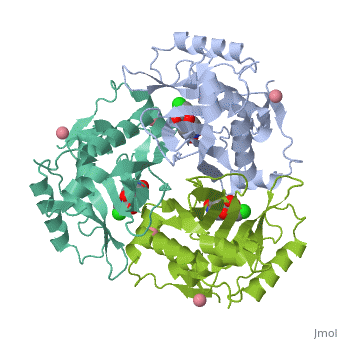
Chloramphenicol acetyltransferase type III (CAT III) is an enzyme which catalyzes the transfer of an acetyl group from acetyl-CoA to hydroxyl groups of chloramphenicol. CAT III is a trimeric protein
Introduction
Reaction of CAT III
In the first step of the reaction, Histidine-156 abstracts a proton from the 3-hydroxyl of chloramphenicol, promoting a nucleophilic attack from the deprotonated oxygen to the thioester bond of the acetyl-CoA. The intermediate produced, 3-acetylchloramphenicol, then rearranges non-enzymatically to 1-acetylchloramphenicol. Regeneration of the 3-hydroxyl allows another CAT III catalyzed nucleophilic attack to another acetyl-CoA and a 1,3-diacetylchloramphenicol product is formed. [1]
Structure
|
References
- ↑ Murray IA, Lewendon A, Williams JA, Cullis PM, Shaw WV, Leslie AG. Alternative binding modes for chloramphenicol and 1-substituted chloramphenicol analogues revealed by site-directed mutagenesis and X-ray crystallography of chloramphenicol acetyltransferase. Biochemistry. 1991 Apr 16;30(15):3763-70. PMID:2015231
| Please do NOT make changes to this Sandbox until after April 23, 2010. Sandboxes 151-200 are reserved until then for use by the Chemistry 307 class at UNBC taught by Prof. Andrea Gorrell. |