This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Interleukin-10
From Proteopedia
| Line 4: | Line 4: | ||
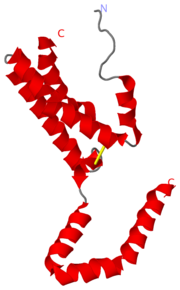
The affinity of IL-10R1 and Il-10R2 are not considered to be dependent on the amino acid sequence but something more complex. There are several homolog IL-10s and mimic IL-10s that are able to bind and signal through the IL-10 receptor complex causing overlaying signals with cIL-10 <ref name="ref1"/>. The completed ternary complex activates the [http://en.wikipedia.org/wiki/JAK-STAT_signaling_pathway JAK/STAT signaling pathway].[[Image:IL10.png|left|thumb|'''Figure 2. Sequential assembly of the ternary complex''' ]] The completion of the ternary complex is dependent on the conformational changes that occur in the N terminus of the helix labelled A. The conformational changes occur when the cIL-10 binds with IL-10R1. There is a positional change of about 1 angstrom at residues between Cys-12 and Leu-46. There are structural features that allow for the large conformational changes, the structural features are a Cys-12 to Cys-108 disulfide bond at the N terminus and Leu-Leu-Leu motifat the C terminus of the AB loop<ref name="ref1"/>. | The affinity of IL-10R1 and Il-10R2 are not considered to be dependent on the amino acid sequence but something more complex. There are several homolog IL-10s and mimic IL-10s that are able to bind and signal through the IL-10 receptor complex causing overlaying signals with cIL-10 <ref name="ref1"/>. The completed ternary complex activates the [http://en.wikipedia.org/wiki/JAK-STAT_signaling_pathway JAK/STAT signaling pathway].[[Image:IL10.png|left|thumb|'''Figure 2. Sequential assembly of the ternary complex''' ]] The completion of the ternary complex is dependent on the conformational changes that occur in the N terminus of the helix labelled A. The conformational changes occur when the cIL-10 binds with IL-10R1. There is a positional change of about 1 angstrom at residues between Cys-12 and Leu-46. There are structural features that allow for the large conformational changes, the structural features are a Cys-12 to Cys-108 disulfide bond at the N terminus and Leu-Leu-Leu motifat the C terminus of the AB loop<ref name="ref1"/>. | ||
<scene name='Sandbox_165/2h24_backbone/1'>2h24 ball and stick</scene> | <scene name='Sandbox_165/2h24_backbone/1'>2h24 ball and stick</scene> | ||
| + | |||
| + | |||
| + | |||
| + | |||
=='''IL-10'''== | =='''IL-10'''== | ||
Revision as of 05:14, 1 April 2010
Contents |
Interleukin-10
2H24 is the PDB id assigned to Interleukin-10 (IL-10) after the determination of the crystal structure. IL-10 is a ternary complex that requires specific assembly for proper function. The IL-10 complex is composed of IL-10,IL-10R1,IL-10R2 (R=receptor). The initial step is the formation of IL-10 and IL-10R1, a binary complex that generates a conformational change. This conformational change is required for IL-10R2 to be able to associate with the binary complex and form the ternary complex [1].
The affinity of IL-10R1 and Il-10R2 are not considered to be dependent on the amino acid sequence but something more complex. There are several homolog IL-10s and mimic IL-10s that are able to bind and signal through the IL-10 receptor complex causing overlaying signals with cIL-10 [1]. The completed ternary complex activates the JAK/STAT signaling pathway. The completion of the ternary complex is dependent on the conformational changes that occur in the N terminus of the helix labelled A. The conformational changes occur when the cIL-10 binds with IL-10R1. There is a positional change of about 1 angstrom at residues between Cys-12 and Leu-46. There are structural features that allow for the large conformational changes, the structural features are a Cys-12 to Cys-108 disulfide bond at the N terminus and Leu-Leu-Leu motifat the C terminus of the AB loop[1].
IL-10
Interleukin-10 is in the class cytokine.[1] Interleukin-10 is a very powerful anti-inflammatory cytokine that is most commonly found to be produce by moncytes [2]. IL-10 has the ability to be a immunosupressive as well as a anti-angiogenic this means that it has the ability both promot and inhibit tumors[3]. There are many types of cytokines that have many funtions such as antiinflammatory cytokines,cytokine synthesis inhibitory factor and proinflammitory cytokines[4].
There are many interleukins that fall within the cytokine class such as IL-1,IL-2,IL-3,IL-4...IL-31,IL-32,IL-33,IL-3 and IL-35.
| |||||||||
| 2h24, resolution 2.00Å () | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Related: | 1inr | ||||||||
| |||||||||
| |||||||||
| |||||||||
| Resources: | FirstGlance, OCA, PDBsum, RCSB | ||||||||
| Coordinates: | save as pdb, mmCIF, xml | ||||||||
Interleukin-10 and Rheumatoid Arthritis
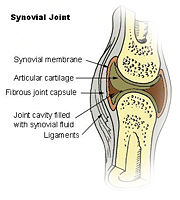
[5]IL-10 has been investigated for its role in patients with Rheumatoid Arthritis (RA) as well as those with Osteoarthritis (OA)[4]. RA is an autoimmune disorder that affects the synovial tissues via chronic synovitis. Chronic synovitis often results in joint destruction due to re-absorption of bone and the distruction of cartilage[6] [4]. IL-10 is found to be spontaneously produce in synovial tissue of patients with RA and OA but not in normal synovial tissue. The variation in secretion of IL-10 is thought to be 75% under the control of the genetics of the individual[6]. When IL-10 is present there is an inhibition of the proinflammatory cytokines, namely TFN-α, IL-1α, IL-1β, IL-6, IL-8 [7].
| Please do NOT make changes to this Sandbox until after April 23, 2010. Sandboxes 151-200 are reserved until then for use by the Chemistry 307 class at UNBC taught by Prof. Andrea Gorrell. |
Proteopedia Page Contributors and Editors (what is this?)
Shelly Huebert, Andrea Gorrell, Alexander Berchansky, David Canner, Jaime Prilusky, Michal Harel