This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
2psm
From Proteopedia
(New page: 200px<br /><applet load="2psm" size="350" color="white" frame="true" align="right" spinBox="true" caption="2psm, resolution 2.19Å" /> '''Crystal structure of...) |
|||
| Line 4: | Line 4: | ||
==Overview== | ==Overview== | ||
| - | Interleukin (IL)-15 is a pleiotropic cytokine that plays a pivotal role in | + | Interleukin (IL)-15 is a pleiotropic cytokine that plays a pivotal role in both innate and adaptive immunity. IL-15 is unique among cytokines due to its participation in a trans signaling mechanism in which IL-15 receptor alpha (IL-15Ralpha) from one subset of cells presents IL-15 to neighboring IL-2Rbeta/gammac-expressing cells. Here we present the crystal structure of IL-15 in complex with the sushi domain of IL-15Ralpha. The structure reveals that the alpha receptor-binding epitope of IL-15 adopts a unique conformation, which, together with amino acid substitutions, permits specific interactions with IL-15Ralpha that account for the exceptionally high affinity of the IL-15.IL-15Ralpha complex. Interestingly, analysis of the topology of IL-15 and IL-15Ralpha at the IL-15.IL-15Ralpha interface suggests that IL-15 should be capable of participating in a cis signaling mechanism similar to that of the related cytokine IL-2. Indeed, we present biochemical data demonstrating that IL-15 is capable of efficiently signaling in cis through IL-15Ralpha and IL-2Rbeta/gammac expressed on the surface of a single cell. Based on our data we propose that cis presentation of IL-15 may be important in certain biological contexts and that flexibility of IL-15Ralpha permits IL-15 and its three receptor components to be assembled identically at the ligand-receptor interface whether IL-15 is presented in cis or trans. Finally, we have gained insights into IL-15.IL-15Ralpha.IL-2Rbeta.gammac quaternary complex assembly through the use of molecular modeling. |
==About this Structure== | ==About this Structure== | ||
| Line 10: | Line 10: | ||
==Reference== | ==Reference== | ||
| - | Crystal | + | Crystal Structure of the interleukin-15.interleukin-15 receptor alpha complex: insights into trans and cis presentation., Olsen SK, Ota N, Kishishita S, Kukimoto-Niino M, Murayama K, Uchiyama H, Toyama M, Terada T, Shirouzu M, Kanagawa O, Yokoyama S, J Biol Chem. 2007 Dec 21;282(51):37191-204. Epub 2007 Oct 18. PMID:[http://ispc.weizmann.ac.il//pmbin/getpm?pmid=17947230 17947230] |
[[Category: Mus musculus]] | [[Category: Mus musculus]] | ||
[[Category: Protein complex]] | [[Category: Protein complex]] | ||
| Line 17: | Line 17: | ||
[[Category: Kukimoto-Niino, M.]] | [[Category: Kukimoto-Niino, M.]] | ||
[[Category: Murayama, K.]] | [[Category: Murayama, K.]] | ||
| - | [[Category: Olsen, S | + | [[Category: Olsen, S K.]] |
[[Category: Ota, N.]] | [[Category: Ota, N.]] | ||
| - | [[Category: RSGI, RIKEN | + | [[Category: RSGI, RIKEN Structural Genomics/Proteomics Initiative.]] |
[[Category: Shirouzu, M.]] | [[Category: Shirouzu, M.]] | ||
[[Category: Terada, T.]] | [[Category: Terada, T.]] | ||
| Line 42: | Line 42: | ||
[[Category: transmembrane]] | [[Category: transmembrane]] | ||
| - | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on | + | ''Page seeded by [http://oca.weizmann.ac.il/oca OCA ] on Thu Feb 21 18:32:40 2008'' |
Revision as of 16:32, 21 February 2008
|
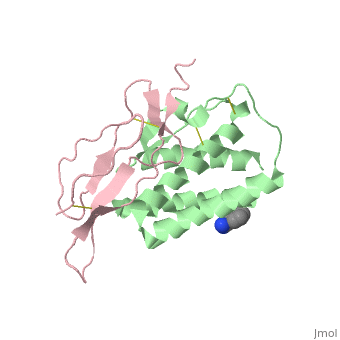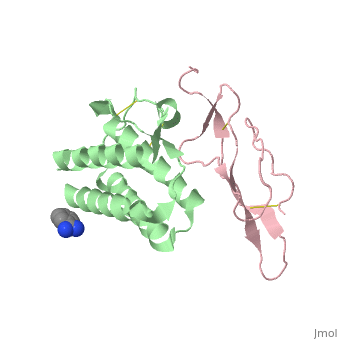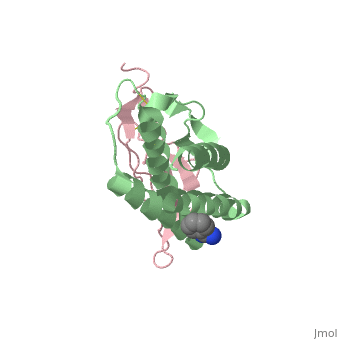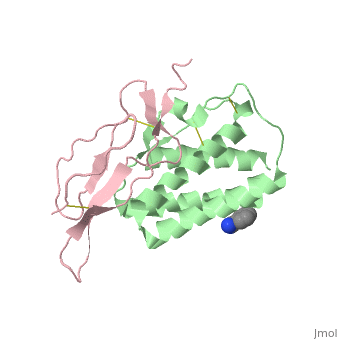
Crystal structure of Interleukin 15 in complex with Interleukin 15 receptor alpha
Overview
Interleukin (IL)-15 is a pleiotropic cytokine that plays a pivotal role in both innate and adaptive immunity. IL-15 is unique among cytokines due to its participation in a trans signaling mechanism in which IL-15 receptor alpha (IL-15Ralpha) from one subset of cells presents IL-15 to neighboring IL-2Rbeta/gammac-expressing cells. Here we present the crystal structure of IL-15 in complex with the sushi domain of IL-15Ralpha. The structure reveals that the alpha receptor-binding epitope of IL-15 adopts a unique conformation, which, together with amino acid substitutions, permits specific interactions with IL-15Ralpha that account for the exceptionally high affinity of the IL-15.IL-15Ralpha complex. Interestingly, analysis of the topology of IL-15 and IL-15Ralpha at the IL-15.IL-15Ralpha interface suggests that IL-15 should be capable of participating in a cis signaling mechanism similar to that of the related cytokine IL-2. Indeed, we present biochemical data demonstrating that IL-15 is capable of efficiently signaling in cis through IL-15Ralpha and IL-2Rbeta/gammac expressed on the surface of a single cell. Based on our data we propose that cis presentation of IL-15 may be important in certain biological contexts and that flexibility of IL-15Ralpha permits IL-15 and its three receptor components to be assembled identically at the ligand-receptor interface whether IL-15 is presented in cis or trans. Finally, we have gained insights into IL-15.IL-15Ralpha.IL-2Rbeta.gammac quaternary complex assembly through the use of molecular modeling.
About this Structure
2PSM is a Protein complex structure of sequences from Mus musculus with as ligand. Full crystallographic information is available from OCA.
Reference
Crystal Structure of the interleukin-15.interleukin-15 receptor alpha complex: insights into trans and cis presentation., Olsen SK, Ota N, Kishishita S, Kukimoto-Niino M, Murayama K, Uchiyama H, Toyama M, Terada T, Shirouzu M, Kanagawa O, Yokoyama S, J Biol Chem. 2007 Dec 21;282(51):37191-204. Epub 2007 Oct 18. PMID:17947230
Page seeded by OCA on Thu Feb 21 18:32:40 2008
Categories: Mus musculus | Protein complex | Kanagawa, O. | Kishishita, S. | Kukimoto-Niino, M. | Murayama, K. | Olsen, S K. | Ota, N. | RSGI, RIKEN Structural Genomics/Proteomics Initiative. | Shirouzu, M. | Terada, T. | Yokoyama, S. | BAM | Alternative splicing | Cytokine | Endoplasmic reticulum | Glycoprotein | Golgi apparatus | Membrane | National project on protein structural and functional analyses | Nppsfa | Nucleus | Phosphorylation | Receptor | Riken structural genomics/proteomics initiative | Rsgi | Secreted | Structural genomics | Sushi | Transmembrane