This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Angiotensin-Converting Enzyme
From Proteopedia
| Line 1: | Line 1: | ||
| - | <StructureSection load='1o8a' size='450' side='right' scene='Angiotensin-Converting_Enzyme/Ace_opening/1' caption=''> | + | <StructureSection load='1o8a' size='450' side='right' scene='Angiotensin-Converting_Enzyme/Ace_opening/1' caption='Human ACE complex with Zn+2 (grey) and Cl- (yellow) ions (PDB code [[1o8a]])'> |
__NOTOC__ | __NOTOC__ | ||
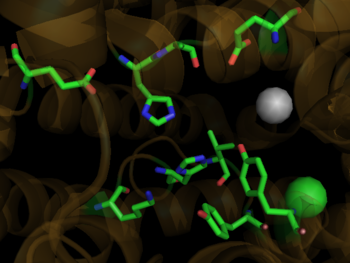
[[Image: 1o8a2.png|320px|left|thumb| Crystal Structure of Human tACE, [[1o8a]]]] | [[Image: 1o8a2.png|320px|left|thumb| Crystal Structure of Human tACE, [[1o8a]]]] | ||
Revision as of 08:19, 3 December 2013
| |||||||||||
3D Structures of Angiotensin-Converting Enzyme
Updated on 03-December-2013
1o8a – hANCE - human
2iul, 2iux, 3nxq – hANCE (mutant)
2c6f – hANCE N domain
2c6n - hANCE N domain+lisinopril
1o86 - hANCE +lisinopril
2ydm - hANCE + captopril analog
3kbh, 3d0g, 3d0h, 3d0i, 2ajf – hANCE 2 fragment+spike glycoprotein
3l3n – hANCE+LISW
3bkk, 3bkl – hANCE+ketone inhibitor
2oc2 - hANCE+phosphinic inhibitor
2xy9, 2xyd - hANCE N domain + phosphinic inhibitor
1uze, 1uzf - hANCE+anti-hypertensive drug
4aph – hANCE + angiotensin II
4apj – hANCE + bradykinin-potentiating peptide B
2xhm – DmANCE+K26 – Drosophila melanogaster
2x8y – DmANCE
2x8z, 2x90, 2x91, 2x92, 2x93, 2x94, 2x95, 2x96, 2x97, 1j36, 1j37, 1j38 – DmANCE+anti-hypertensive drug
3zqz – DmANCE + captopril analog
4aa1 – DmANCE + angiotensin II
4aa2, 4asr – DmANCE + bradykinin-potentiating peptide B
4asq – DmANCE + bradykinin peptide
Additional Resources
For Additional Information, see: Hypertension & Congestive Heart Failure
References
- ↑ Skeggs, L. T., Dorer, F. E., Kahn, J. R., Lentz, K. E., Levin, M. (1981) Experimental renal hypertension: the discovery of the Renin-Angiotensin system. Soffer, R. eds. Biochemical Regulation of Blood Pressure ,3-38 John Wiley & Sons, Inc. Hoboken.
- ↑ Hoogwerf BJ, Young JB. The HOPE study. Ramipril lowered cardiovascular risk, but vitamin E did not. Cleve Clin J Med. 2000 Apr;67(4):287-93. PMID:10780101
- ↑ 3.0 3.1 3.2 Ferrario CM. Role of angiotensin II in cardiovascular disease therapeutic implications of more than a century of research. J Renin Angiotensin Aldosterone Syst. 2006 Mar;7(1):3-14. PMID:17083068
- ↑ Spyroulias GA, Nikolakopoulou P, Tzakos A, Gerothanassis IP, Magafa V, Manessi-Zoupa E, Cordopatis P. Comparison of the solution structures of angiotensin I & II. Implication for structure-function relationship. Eur J Biochem. 2003 May;270(10):2163-73. PMID:12752436
- ↑ 5.0 5.1 Brew K. Structure of human ACE gives new insights into inhibitor binding and design. Trends Pharmacol Sci. 2003 Aug;24(8):391-4. PMID:12915047
- ↑ 6.0 6.1 Sturrock ED, Natesh R, van Rooyen JM, Acharya KR. Structure of angiotensin I-converting enzyme. Cell Mol Life Sci. 2004 Nov;61(21):2677-86. PMID:15549168 doi:10.1007/s00018-004-4239-0
- ↑ 7.0 7.1 7.2 Weir MR. Effects of renin-angiotensin system inhibition on end-organ protection: can we do better? Clin Ther. 2007 Sep;29(9):1803-24. PMID:18035185 doi:10.1016/j.clinthera.2007.09.019
- ↑ Henriksen EJ, Jacob S. Modulation of metabolic control by angiotensin converting enzyme (ACE) inhibition. J Cell Physiol. 2003 Jul;196(1):171-9. PMID:12767053 doi:10.1002/jcp.10294
- ↑ Cole J, Ertoy D, Bernstein KE. Insights derived from ACE knockout mice. J Renin Angiotensin Aldosterone Syst. 2000 Jun;1(2):137-41. PMID:11967804
- ↑ Junot C, Gonzales MF, Ezan E, Cotton J, Vazeux G, Michaud A, Azizi M, Vassiliou S, Yiotakis A, Corvol P, Dive V. RXP 407, a selective inhibitor of the N-domain of angiotensin I-converting enzyme, blocks in vivo the degradation of hemoregulatory peptide acetyl-Ser-Asp-Lys-Pro with no effect on angiotensin I hydrolysis. J Pharmacol Exp Ther. 2001 May;297(2):606-11. PMID:11303049
- ↑ 11.0 11.1 11.2 11.3 11.4 11.5 Natesh R, Schwager SL, Sturrock ED, Acharya KR. Crystal structure of the human angiotensin-converting enzyme-lisinopril complex. Nature. 2003 Jan 30;421(6922):551-4. Epub 2003 Jan 19. PMID:12540854 doi:http://dx.doi.org/10.1038/nature01370
- ↑ Hangauer DG, Monzingo AF, Matthews BW. An interactive computer graphics study of thermolysin-catalyzed peptide cleavage and inhibition by N-carboxymethyl dipeptides. Biochemistry. 1984 Nov 20;23(24):5730-41. PMID:6525336
- ↑ Jaspard E, Alhenc-Gelas F. Catalytic properties of the two active sites of angiotensin I-converting enzyme on the cell surface. Biochem Biophys Res Commun. 1995 Jun 15;211(2):528-34. PMID:7794265
- ↑ http://www.yourlawyer.com/topics/overview/ace_inhibitors
- ↑ Natesh R, Schwager SL, Evans HR, Sturrock ED, Acharya KR. Structural details on the binding of antihypertensive drugs captopril and enalaprilat to human testicular angiotensin I-converting enzyme. Biochemistry. 2004 Jul 13;43(27):8718-24. PMID:15236580 doi:10.1021/bi049480n
- ↑ 16.0 16.1 Li F. Structural analysis of major species barriers between humans and palm civets for severe acute respiratory syndrome coronavirus infections. J Virol. 2008 Jul;82(14):6984-91. Epub 2008 Apr 30. PMID:18448527 doi:10.1128/JVI.00442-08
Proteopedia Page Contributors and Editors (what is this?)
David Canner, Michal Harel, Alexander Berchansky, Cristina Murga