This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox bcce11
From Proteopedia
(Difference between revisions)
(New page: Human P-glycoprotein Structure ==Your Heading Here (maybe something like 'Structure')== <StructureSection load='1stp' size='340' side='right' caption='Caption for this structure' scene=''>...) |
|||
| (8 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | Human P-glycoprotein | + | ==Human P-glycoprotein== |
| - | + | ||
| - | <StructureSection load=' | + | <StructureSection load='3G61' size='340' side='right' caption='Caption for this structure' scene=''> |
| - | + | ||
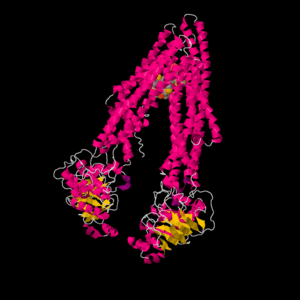
| - | + | Human P-glycoprotein (ABCB1, P-gp) is a membrane protein present primarily in sanctuary sites, such as the blood-brain barrier, blood-testes barrier, placenta, and gut. The exact mechanism and quantity of molecules bound to P-gp during one transport cycle has not been confirmed experimentally, however recent advances in structural information have begun to reveal a more detailed image of subtrate binding to P-gp <ref>PMID:19325113</ref> | |
| + | |||
| + | [[Image:Jsmol.gif|300 px|]] | ||
| + | (Image showing inhibitor bound to P-gp.) | ||
| + | |||
| + | <scene name='59/596453/Ligand/1'>Ligand bound to P-gp</scene> | ||
| + | <scene name='59/596453/Ligand/2'>Ligand zoom</scene> | ||
| + | <scene name='59/596453/Ligand/3'>No rotation</scene> | ||
== Function == | == Function == | ||
Current revision
Human P-glycoprotein
| |||||||||||
References
- ↑ Aller SG, Yu J, Ward A, Weng Y, Chittaboina S, Zhuo R, Harrell PM, Trinh YT, Zhang Q, Urbatsch IL, Chang G. Structure of P-glycoprotein reveals a molecular basis for poly-specific drug binding. Science. 2009 Mar 27;323(5922):1718-22. PMID:19325113 doi:323/5922/1718