This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1061
From Proteopedia
(Difference between revisions)
| Line 14: | Line 14: | ||
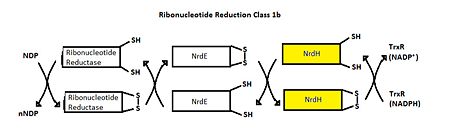
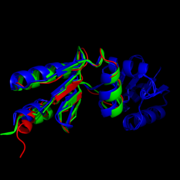
The <scene name='69/694228/Nrdh_structure/1'>MtNrdH structure</scene> determined by x-ray crystallography has 79 residues in a single polypeptide chain. The active site (shown in green) is dominated by a <scene name='69/694228/Nrdh_structure/3'>disulfide bond</scene> between Cys-11 and Cys-14, which serves as the site of reduction by thioredoxin reductase. <ref name="Swastik" /> | The <scene name='69/694228/Nrdh_structure/1'>MtNrdH structure</scene> determined by x-ray crystallography has 79 residues in a single polypeptide chain. The active site (shown in green) is dominated by a <scene name='69/694228/Nrdh_structure/3'>disulfide bond</scene> between Cys-11 and Cys-14, which serves as the site of reduction by thioredoxin reductase. <ref name="Swastik" /> | ||
| - | Many thioredoxin-like proteins have a similar active site region, which includes the <scene name='69/694228/Nrdh_structure/ | + | Many thioredoxin-like proteins have a similar active site region, which includes the |
| + | <scene name='69/694228/Nrdh_structure/6'>thioredoxin fold</scene>, a large turn in the protein structure right before the disulfide bond. The residues directly following the fold, <scene name='69/694228/Nrdh_structure/5'>CVQC</scene>, are the most highly conserved of all areas of the protein across multiple species. | ||
[[Image:Weblogocvqc.png|thumb|center|upright=2.5|Weblogo diagram showing highly conserved CVQC region of NrdH in five separate protein structures from ''Nocardiaseriolae'', ''E. coli'', ''Cornebacterium Ammoniagenes'', and ''Mycobacterium Tuberculosis''.<ref name="weblogo">Crooks GE, Hon G, Chandonia JM, Brenner SE WebLogo: A sequence logo generator, | [[Image:Weblogocvqc.png|thumb|center|upright=2.5|Weblogo diagram showing highly conserved CVQC region of NrdH in five separate protein structures from ''Nocardiaseriolae'', ''E. coli'', ''Cornebacterium Ammoniagenes'', and ''Mycobacterium Tuberculosis''.<ref name="weblogo">Crooks GE, Hon G, Chandonia JM, Brenner SE WebLogo: A sequence logo generator, | ||
Genome Research, 14:1188-1190, (2004)</ref>]] | Genome Research, 14:1188-1190, (2004)</ref>]] | ||
Revision as of 00:28, 22 April 2015
| This Sandbox is Reserved from 02/09/2015, through 05/31/2016 for use in the course "CH462: Biochemistry 2" taught by Geoffrey C. Hoops at the Butler University. This reservation includes Sandbox Reserved 1051 through Sandbox Reserved 1080. |
To get started:
More help: Help:Editing |
Structure of Mycobacterium Tuberculosis NrdH
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 1.7 Swastik, Phulera and Mande, Shekhar C. (2013) The Crystal Structure of Mycobacterium tuberculosis NrdH at 0.87Å Suggests a Possible Mode of Its Activity. Biochemistry 52, 4056-4065.
- ↑ 2.0 2.1 "Tuberculosis." Media Centre. World Health Organization, Web. 16 Mar. 2015. Media Centre. <http://www.who.int/mediacentre/factsheets/fs104/en/>.
- ↑ 3.0 3.1 Crooks GE, Hon G, Chandonia JM, Brenner SE WebLogo: A sequence logo generator, Genome Research, 14:1188-1190, (2004)
- ↑ 4.0 4.1 The PyMOL Molecular Graphics System, Version 1.7.4 Schrödinger, LLC.
- ↑ 5.0 5.1 Kolberg, M., et al. (2004) Structure, function, and mechanism of ribonucleotide reductases. Biochim. Biophys. Acta 1699 (1−2), 1−34.
- ↑ Nelson, David L., and Michael M. Cox. Lehninger Principles of Biochemistry. 5th ed. New York: W.H. Freeman, 2008. 888-889.
- ↑ Makhlynets, O., Boal, A. K., Rhodes, D. V., Kitten, T., Rosenzweig, A. C., & Stubbe, J. (2014). Streptococcus sanguinis Class Ib Ribonucleotide Reductase: HIGH ACTIVITY WITH BOTH IRON AND MANGANESE COFACTORS AND STRUCTURAL INSIGHTS. The Journal of Biological Chemistry, 289(9), 6259–6272. doi:10.1074/jbc.M113.533554.
- ↑ Mowa, M. B., et al. (2009) Function and regulation of class I ribonucleotide reductase-encoding genes in mycobacteria. J. Bacteriol. 191 (3), 985−995