This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Cathepsin
From Proteopedia
(Difference between revisions)
| Line 21: | Line 21: | ||
== Structural highlights == | == Structural highlights == | ||
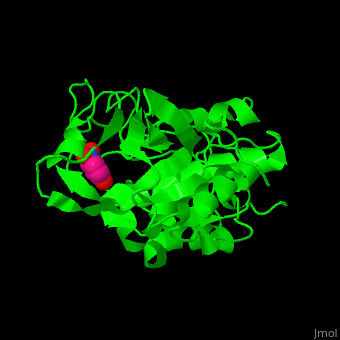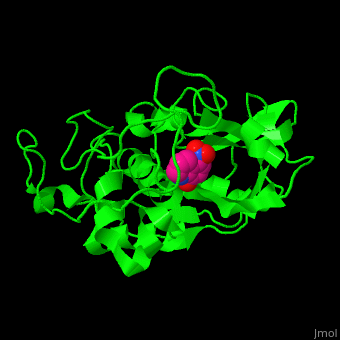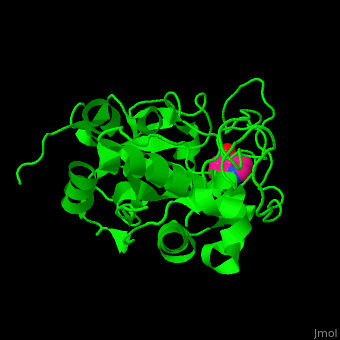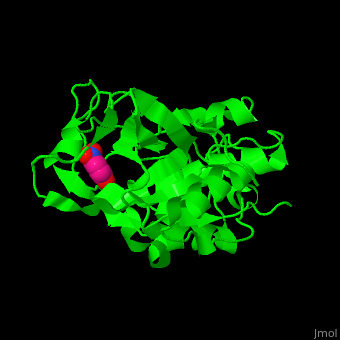
| - | CTS B is a cysteine protease. The <scene name='42/421669/Cv/4'>active site</scene> contains a <scene name='42/421669/Cv/ | + | CTS B is a cysteine protease. The <scene name='42/421669/Cv/4'>active site</scene> contains a <scene name='42/421669/Cv/9'>C29 residue from the N-terminal domain and H199 residue from the C-terminal domain</scene> (in cyan).<ref>PMID:21598397</ref> |
</StructureSection> | </StructureSection> | ||
== 3D Structures of Cathepsin == | == 3D Structures of Cathepsin == | ||
Revision as of 09:55, 29 November 2015
| |||||||||||
3D Structures of Cathepsin
Updated on 29-November-2015
Cathepsin-C mature see Dipeptidyl peptidase-I
References
- ↑ Nomura T, Katunuma N. Involvement of cathepsins in the invasion, metastasis and proliferation of cancer cells. J Med Invest. 2005 Feb;52(1-2):1-9. PMID:15751268
- ↑ Mirkovic B, Renko M, Turk S, Sosic I, Jevnikar Z, Obermajer N, Turk D, Gobec S, Kos J. Novel Mechanism of Cathepsin B Inhibition by Antibiotic Nitroxoline and Related Compounds. ChemMedChem. 2011 May 20. doi: 10.1002/cmdc.201100098. PMID:21598397 doi:10.1002/cmdc.201100098