This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1137
From Proteopedia
(Difference between revisions)
| Line 23: | Line 23: | ||
---- | ---- | ||
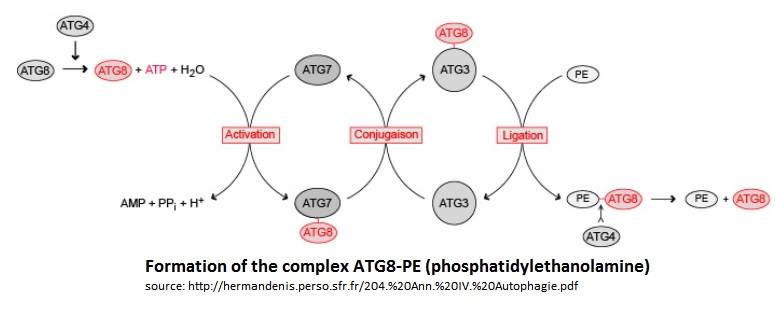
| - | Atg3 is a protein composed of 314 aminoacids with an alpha/beta-fold with a core region topologically similar to E2 enzymes. The core region has two regions: | + | Atg3 is a protein composed of 314 aminoacids with an alpha/beta-fold with a core region topologically similar to [https://en.wikipedia.org/wiki/Ubiquitin-conjugating_enzyme E2 enzymes] . The core region has two regions: |
→ the first region has 80 residues and has a random coil structure in solution, this region is responsible for the Atg7 interaction which is an E1-like enzyme. | → the first region has 80 residues and has a random coil structure in solution, this region is responsible for the Atg7 interaction which is an E1-like enzyme. | ||
→ The second is an alpha-helical structure which protrudes from the core region. This alpha-helical structure is responsible for binding Atg8. | → The second is an alpha-helical structure which protrudes from the core region. This alpha-helical structure is responsible for binding Atg8. | ||
| - | Moreover some researches | + | Moreover some researches indicates that the catalytic cysteine of Atg3 is a possible binding site for a phosphate of phosphatidylethanolamine. |
===Atg8 protein=== | ===Atg8 protein=== | ||
---- | ---- | ||
| - | Atg8 is a protein of 117 aminoacids with a molecular | + | Atg8 is a protein of 117 aminoacids with a molecular wieght of 13,6kDa. This molecule is composed of 5- stranded β-sheet. Those β-sheet are enclosed by two α-helices on each sides. This conformation leaves accessible a conserved GABARAP domain, this protein has been originally identified as a binding partner of a GABAA receptor subunit. [https://en.wikipedia.org/wiki/GABARAP GABARAP]. Even if the sequences between Atg8 and ubiquitin are not similars, the crystal structure reveals a conserved ubiquitine-like fold. Atg8 belongs to the ATG family but it differs from the other members of the family because the α2 helix-terminating proline 26 was substituted by a lysine. |
===Atg8/Atg3 Complex=== | ===Atg8/Atg3 Complex=== | ||
---- | ---- | ||
Revision as of 19:53, 29 January 2016
| This Sandbox is Reserved from 15/12/2015, through 15/06/2016 for use in the course "Structural Biology" taught by Bruno Kieffer at the University of Strasbourg, ESBS. This reservation includes Sandbox Reserved 1120 through Sandbox Reserved 1159. |
To get started:
More help: Help:Editing |
Plasmodium falciparum Atg8 in complex with Plasmodium falciparum Atg3 peptide
| |||||||||||
References
http://hermandenis.perso.sfr.fr/204.%20Ann.%20IV.%20Autophagie.pdf