This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
SAM-dependent methyltransferase
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
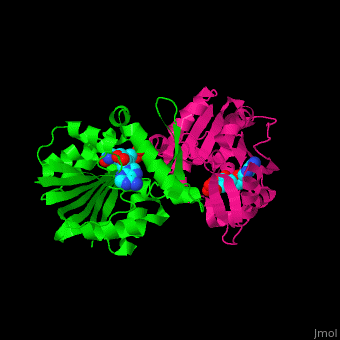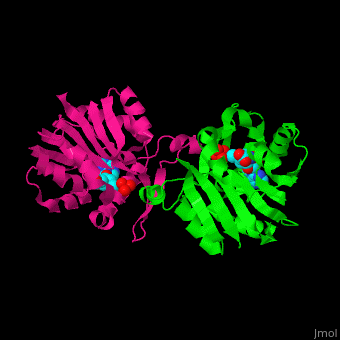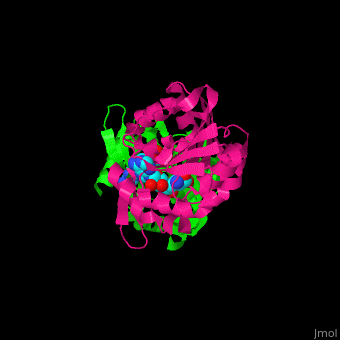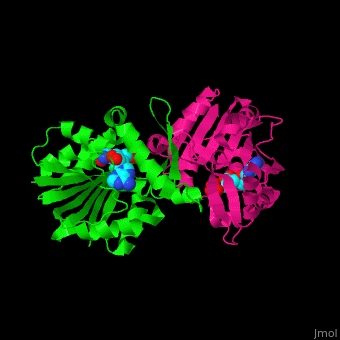
| - | + | <StructureSection load='3ou6' size='350' side='right' scene='' caption='SAM-dependent methyltransferase dimer complex with S-adenosyl-L-homocysteine and sulfate [[3ou6]]'> | |
== Function == | == Function == | ||
'''SAM-dependent methyltransferase''' (SDM) utilizes the methyl donor S-adenosyl-L-methionine (SAM) as a cofactor to methylate proteins, small molecules, lipids and nucleic acids. SAM forms S-adenosyl-L-homocysteine (SAH) upon demethylation. About 120 members of the SDM family have been identified. They differ in their substrate specificity and the atom targeted for methylation (N, O, C, S)<ref>PMID:23180741</ref>. | '''SAM-dependent methyltransferase''' (SDM) utilizes the methyl donor S-adenosyl-L-methionine (SAM) as a cofactor to methylate proteins, small molecules, lipids and nucleic acids. SAM forms S-adenosyl-L-homocysteine (SAH) upon demethylation. About 120 members of the SDM family have been identified. They differ in their substrate specificity and the atom targeted for methylation (N, O, C, S)<ref>PMID:23180741</ref>. | ||
| + | |||
| + | == Structural highlights == | ||
| + | The core of the SDM fold contains alternating β strands and α helices. SDM active site is located between the 2 monomers<ref>PMID:20876132</ref>. | ||
| + | |||
<StructureSection load='1af7' size='350' side='right' scene='47/479962/Cv/1' caption='Chemotaxis receptor methyltransferase CheR complex with S-adenosyl-L-homocysteine, [[1af7]]'> | <StructureSection load='1af7' size='350' side='right' scene='47/479962/Cv/1' caption='Chemotaxis receptor methyltransferase CheR complex with S-adenosyl-L-homocysteine, [[1af7]]'> | ||
For '''Chemotaxis receptor methyltransferase CheR''' see details in [[Molecular Playgroun]d/CheR]].<ref>PMID:9628482</ref> | For '''Chemotaxis receptor methyltransferase CheR''' see details in [[Molecular Playgroun]d/CheR]].<ref>PMID:9628482</ref> | ||
| - | + | <scene name='47/479962/Cv/2'>S-adenosyl-L-homocysteine binding site</scene> of Chemotaxis receptor methyltransferase CheR ([[1af7]]).<ref>PMID:9115443</ref> | |
*SEE ALSO [[Chemotaxis protein]]. | *SEE ALSO [[Chemotaxis protein]]. | ||
</StructureSection> | </StructureSection> | ||
| - | |||
| - | == Structural highlights == | ||
| - | The core of the SDM fold contains alternating β strands and α helices. | ||
==3D structures of SAM-dependent methyltrasferase== | ==3D structures of SAM-dependent methyltrasferase== | ||
Revision as of 08:10, 23 August 2016
| |||||||||||
3D structures of SAM-dependent methyltrasferase
Updated on 23-August-2016
| |||||||||||
References
- ↑ Struck AW, Thompson ML, Wong LS, Micklefield J. S-adenosyl-methionine-dependent methyltransferases: highly versatile enzymes in biocatalysis, biosynthesis and other biotechnological applications. Chembiochem. 2012 Dec 21;13(18):2642-55. doi: 10.1002/cbic.201200556. Epub 2012, Nov 23. PMID:23180741 doi:http://dx.doi.org/10.1002/cbic.201200556
- ↑ Lee JH, Bae B, Kuemin M, Circello BT, Metcalf WW, Nair SK, van der Donk WA. Characterization and structure of DhpI, a phosphonate O-methyltransferase involved in dehydrophos biosynthesis. Proc Natl Acad Sci U S A. 2010 Oct 12;107(41):17557-62. Epub 2010 Sep 27. PMID:20876132 doi:10.1073/pnas.1006848107
- ↑ Djordjevic S, Stock AM. Chemotaxis receptor recognition by protein methyltransferase CheR. Nat Struct Biol. 1998 Jun;5(6):446-50. PMID:9628482
- ↑ Djordjevic S, Stock AM. Crystal structure of the chemotaxis receptor methyltransferase CheR suggests a conserved structural motif for binding S-adenosylmethionine. Structure. 1997 Apr 15;5(4):545-58. PMID:9115443
- ↑ Djordjevic S, Stock AM. Chemotaxis receptor recognition by protein methyltransferase CheR. Nat Struct Biol. 1998 Jun;5(6):446-50. PMID:9628482
- ↑ Djordjevic S, Stock AM. Crystal structure of the chemotaxis receptor methyltransferase CheR suggests a conserved structural motif for binding S-adenosylmethionine. Structure. 1997 Apr 15;5(4):545-58. PMID:9115443