This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Tissue factor pathway inhibitor
From Proteopedia
| Line 1: | Line 1: | ||
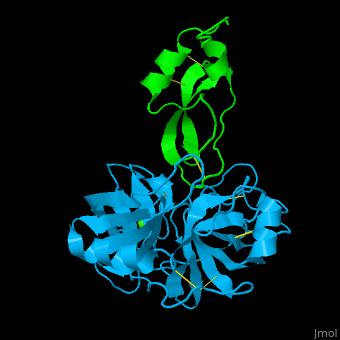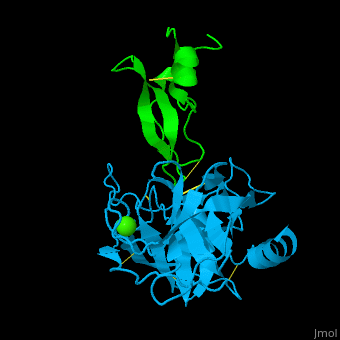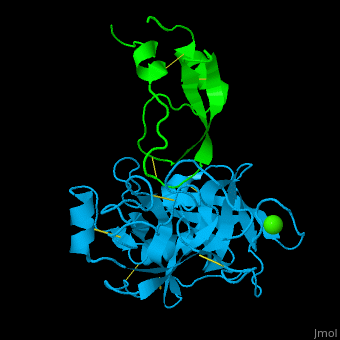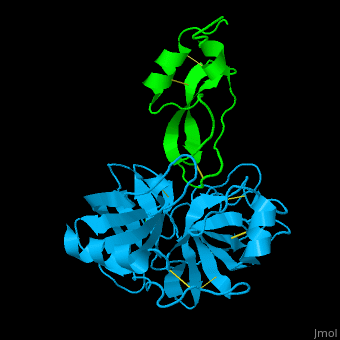
| - | <StructureSection load='1zr0' size='450' side='right' caption='Human tissue factor pathway inhibitor 2 | + | <StructureSection load='1zr0' size='450' side='right' caption='Human tissue factor pathway inhibitor 2 Kunitz domain I (green) complex with trypsin (cyan) and Ca+2 ion (PDB code [[1zr0]])' scene='70/707370/Cv/1'> |
== Function == | == Function == | ||
'''Tissue factor pathway inhibitor''' (TFPI) is a protease inhibitor which inhibits coagulation factor Xa and VIIa<ref>PMID:9112630</ref>. TFPI contains three Kunitz domains. The Kunitz domain is disulfide-rich and is arranged to form a twisted two-stranded antiparallel β sheet followed by an α helix. Kunitz I and II domains inhibit VIIa coagulation factor while Kunitz II domain inhibits Xa. Kunitz III domains probably involved in the interactions with lipoproteins. | '''Tissue factor pathway inhibitor''' (TFPI) is a protease inhibitor which inhibits coagulation factor Xa and VIIa<ref>PMID:9112630</ref>. TFPI contains three Kunitz domains. The Kunitz domain is disulfide-rich and is arranged to form a twisted two-stranded antiparallel β sheet followed by an α helix. Kunitz I and II domains inhibit VIIa coagulation factor while Kunitz II domain inhibits Xa. Kunitz III domains probably involved in the interactions with lipoproteins. | ||
| Line 6: | Line 6: | ||
The expression of hTFPI-2 in tumors is inversely related to their malignancy<ref>PMID:18000791</ref>. TFPI inhibitors are investigated as therapeutic agents agains hemophilia<ref>PMID:27207418</ref>. | The expression of hTFPI-2 in tumors is inversely related to their malignancy<ref>PMID:18000791</ref>. TFPI inhibitors are investigated as therapeutic agents agains hemophilia<ref>PMID:27207418</ref>. | ||
== Structural highlights == | == Structural highlights == | ||
| - | In the complex of TFPI-2 and trypsin, hydrophobic residues of TFPI-2 interact with hydrophobic patch of trypsin. An Arg residue which is the P1 residue of TFPI-2 interacts with Asp - the S1 residue of trypsin<ref>PMID:15932872</ref>. | + | In the complex of TFPI-2 and trypsin, <scene name='70/707370/Cv/2'>hydrophobic residues of TFPI-2 interact with hydrophobic patch of trypsin</scene>. An <scene name='70/707370/Cv/4'>Arg residue which is the P1 residue of TFPI-2 interacts with Asp - the S1 residue of trypsin</scene><ref>PMID:15932872</ref>. |
</StructureSection> | </StructureSection> | ||
Revision as of 07:50, 15 September 2016
| |||||||||||
3D Structures of tissue factor pathway inhibitor
Updated on 15-September-2016
1adz – hTFPI-1 kunitz domain II (mutant) – human - NMR
1irh – hTFPI-1 kunitz domain III - NMR
1tfx – hTFPI-1 kunitz domain II + trypsin
1zr0 – hTFPI-2 kunitz domain I + trypsin
4dtg – hTFPI-1 kunitz domain II + antibody
4bqd – hTFPI-1 kunitz domain I + peptide
References
- ↑ Kato H. Tissue factor pathway inhibitor; its structure, function and clinical significance. Pol J Pharmacol. 1996 Jan-Feb;48(1):67-72. PMID:9112630
- ↑ Sierko E, Wojtukiewicz MZ, Kisiel W. The role of tissue factor pathway inhibitor-2 in cancer biology. Semin Thromb Hemost. 2007 Oct;33(7):653-9. PMID:18000791 doi:http://dx.doi.org/10.1055/s-2007-991532
- ↑ Peterson JA, Maroney SA, Mast AE. Targeting TFPI for hemophilia treatment. Thromb Res. 2016 May;141 Suppl 2:S28-30. doi: 10.1016/S0049-3848(16)30359-0. PMID:27207418 doi:http://dx.doi.org/10.1016/S0049-3848(16)30359-0
- ↑ Schmidt AE, Chand HS, Cascio D, Kisiel W, Bajaj SP. Crystal structure of Kunitz domain 1 (KD1) of tissue factor pathway inhibitor-2 in complex with trypsin. Implications for KD1 specificity of inhibition. J Biol Chem. 2005 Jul 29;280(30):27832-8. Epub 2005 Jun 2. PMID:15932872 doi:10.1074/jbc.M504105200