This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Tumor necrosis factor receptor
From Proteopedia
(Difference between revisions)
| Line 1: | Line 1: | ||
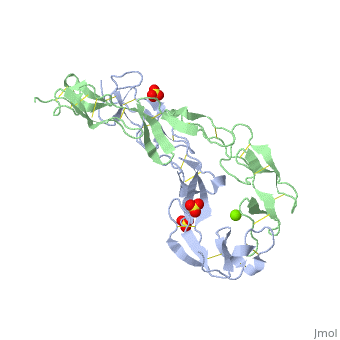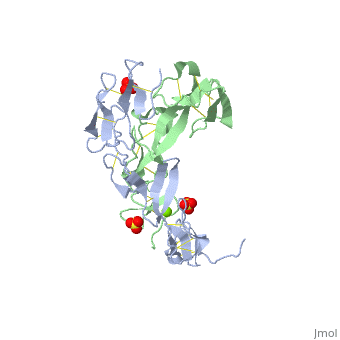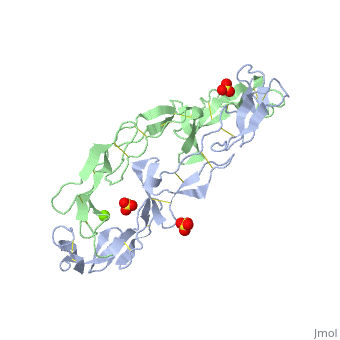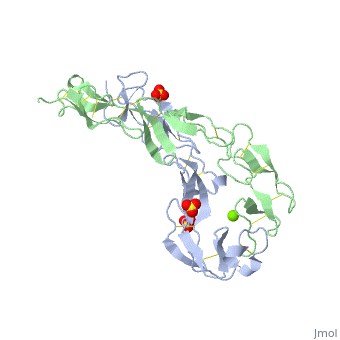
| - | <StructureSection load=' | + | <StructureSection load='1ext' size='340' side='right' caption='Structure of human TNFRSF-18 extracellular domain (PDB code [[2q1m]]).' scene=''> |
== Function == | == Function == | ||
| - | '''Tumor necrosis factor receptor''' (TNFR) or '''death receptor''' is a trimeric cytokine receptor which binds TNF<ref>PMID:10358762</ref>. TNFR family contains several members and superfamily (TNFRSF) members. | + | '''Tumor necrosis factor receptor''' (TNFR) or '''death receptor''' is a trimeric cytokine receptor which binds TNF<ref>PMID:10358762</ref>. TNFR family contains several members and superfamily (TNFRSF) members. <br /> |
* '''TNFRSF 1''' is called '''Lymphotoxin-α or TNF-β''';<br /> | * '''TNFRSF 1''' is called '''Lymphotoxin-α or TNF-β''';<br /> | ||
* '''TNFRSF 3''' is called '''TNFR-III''';<br /> | * '''TNFRSF 3''' is called '''TNFR-III''';<br /> | ||
| Line 23: | Line 23: | ||
* '''TNFRSF 18''' is called '''GITRL''';<br /> | * '''TNFRSF 18''' is called '''GITRL''';<br /> | ||
* '''TNFRSF 21''' is called '''Dr6''';<br /> | * '''TNFRSF 21''' is called '''Dr6''';<br /> | ||
| - | |||
| - | |||
| - | == Disease == | ||
== Relevance == | == Relevance == | ||
| + | TRAPS - a condition characterized by recurrent episodes of fever is associated with TNFR<ref>PMID:24609716</ref>. | ||
== Structural highlights == | == Structural highlights == | ||
| + | The extracellular domain of TNFR contains 2 to 6 cysteine-rich domains (CRD). The CRD domains are ca. 40 amino-acid long and contain 4-6 cysteine residues. The CRD are involved in binding of TNF. | ||
</StructureSection> | </StructureSection> | ||
Revision as of 09:23, 28 September 2016
| |||||||||||
3D structures of tumor necrosis factor receptor
Updated on 28-September-2016
References
- ↑ Wallach D, Varfolomeev EE, Malinin NL, Goltsev YV, Kovalenko AV, Boldin MP. Tumor necrosis factor receptor and Fas signaling mechanisms. Annu Rev Immunol. 1999;17:331-67. PMID:10358762 doi:http://dx.doi.org/10.1146/annurev.immunol.17.1.331
- ↑ Lopalco G, Rigante D, Vitale A, Frediani B, Iannone F, Cantarini L. Tumor necrosis factor receptor-associated periodic syndrome managed with the couple canakinumab-alendronate. Clin Rheumatol. 2015 Apr;34(4):807-9. doi: 10.1007/s10067-014-2556-8. Epub 2014, Mar 11. PMID:24609716 doi:http://dx.doi.org/10.1007/s10067-014-2556-8