This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Ubiquitin
From Proteopedia
(Difference between revisions)
| Line 2: | Line 2: | ||
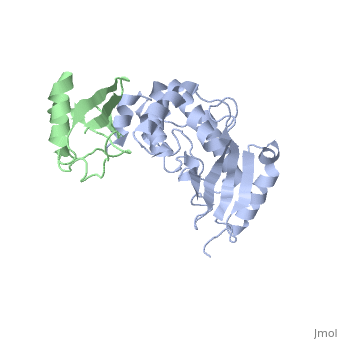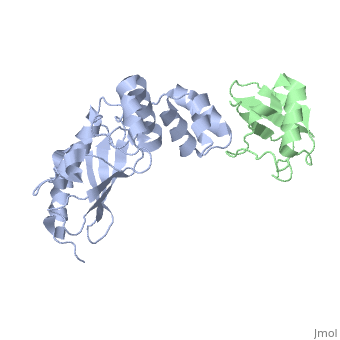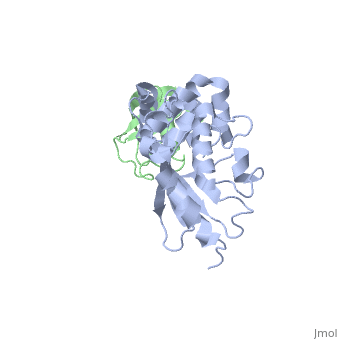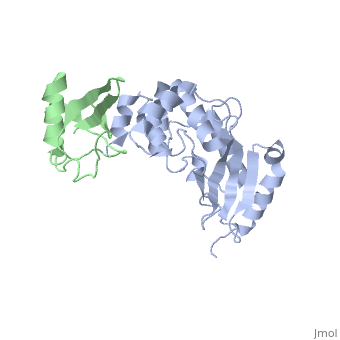
<StructureSection load='3k9p' size='340' side='right' caption='Human ubiquitin (green) complex with ubiquitin-conjugating enzyme E2 (grey), [[3k9p]]' scene='' > | <StructureSection load='3k9p' size='340' side='right' caption='Human ubiquitin (green) complex with ubiquitin-conjugating enzyme E2 (grey), [[3k9p]]' scene='' > | ||
| - | + | -- Function == | |
| - | + | [[Ubiquitin]] (UBB) is found in almost all cells. It binds to proteins tagging them for destruction in the proteasome. UBB is activated by the UBB-activating enzymes E1, E2 and E3. UBB+1 is a frameshifted mutant of UBB observed in several diseases. A dimer of UBB (DiUBB) is formed by linkage of K48 to the C-terminus of a second UBB molecule. At least 4 UBB molecules are needed to tag a protein for the proteasome<ref>PMID:9759494</ref>. For details see<br /> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | [[Ubiquitin]] (UBB) is found in almost all cells. It binds to proteins tagging them for destruction in the proteasome. UBB is activated by the UBB-activating enzymes E1, E2 and E3. UBB+1 is a frameshifted mutant of UBB observed in several diseases. A dimer of UBB (DiUBB) is formed by linkage of K48 to the C-terminus of a second UBB molecule. At least 4 UBB molecules are needed to tag a protein for the proteasome. For details see<br /> | + | |
* [[Ubiquitin Structure & Function]]<br /> | * [[Ubiquitin Structure & Function]]<br /> | ||
* [[Ubiquitin salt bridge discussion]]<br /> | * [[Ubiquitin salt bridge discussion]]<br /> | ||
| Line 31: | Line 10: | ||
==Additional Resources== | ==Additional Resources== | ||
See: [[Ubiquitin Structure & Function]] for additional information. | See: [[Ubiquitin Structure & Function]] for additional information. | ||
| + | |||
| + | == Disease == | ||
| + | The UBB-proteasome system deregulation has been implicated in the pathogenesis of many neurodegenerative disorders like Alzheimer's disease, Parkinson disease, Huntington disease, Prion-like lethal disorders and in genetic diseases like cystic fibrosis, angelman's syndrome, Liddle syndrome and many cancers<ref>PMID:18937370</ref>. | ||
</StructureSection> | </StructureSection> | ||
== 3D Structures of Ubiquitin == | == 3D Structures of Ubiquitin == | ||
| Line 186: | Line 168: | ||
**[[3n32]] – hUBB+Pt<br /> | **[[3n32]] – hUBB+Pt<br /> | ||
}} | }} | ||
| + | ==References== | ||
| + | <references /> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Revision as of 09:56, 29 November 2016
Image:1d3z.png
NMR Structure of Ubiquitin, 1d3z
| |||||||||||
3D Structures of Ubiquitin
Updated on 29-November-2016
References
- ↑ Hershko A, Ciechanover A. The ubiquitin system. Annu Rev Biochem. 1998;67:425-79. PMID:9759494 doi:http://dx.doi.org/10.1146/annurev.biochem.67.1.425
- ↑ Paul S. Dysfunction of the ubiquitin-proteasome system in multiple disease conditions: therapeutic approaches. Bioessays. 2008 Nov;30(11-12):1172-84. doi: 10.1002/bies.20852. PMID:18937370 doi:http://dx.doi.org/10.1002/bies.20852
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Joel L. Sussman, David Canner, Jaime Prilusky