This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Clp Protease
From Proteopedia
(Difference between revisions)
| Line 11: | Line 11: | ||
== Structural highlights == | == Structural highlights == | ||
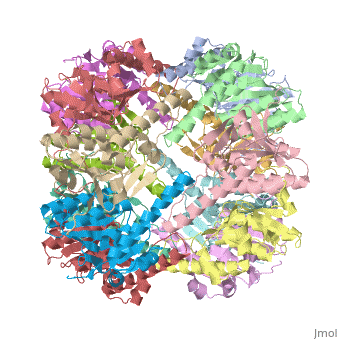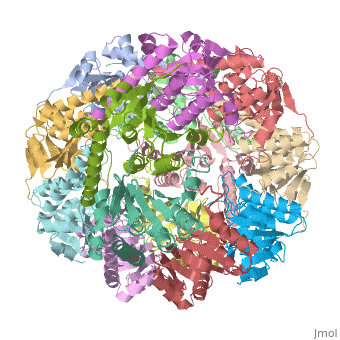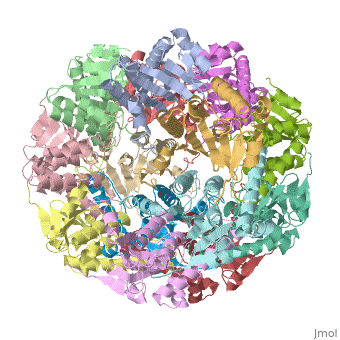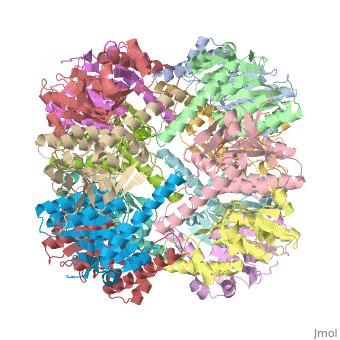
| - | CLP is a heterodimer containing an ATP-binding regulatory subunit A '''ClpA''' and catalytic subunit P '''ClpP'''. The proteolytic complex is composed of 2 heptameric rings of ClpP flanked by hexameric rings of ClpA. For full proteolytic activity ClpP must associate with one of two related ATPase subunits: ClpA and [[ClpX]]. | + | CLP is a heterodimer containing an ATP-binding regulatory subunit A '''ClpA''' or Hsp100 in [[Heat Shock Proteins]] and catalytic subunit P '''ClpP'''. The proteolytic complex is composed of 2 heptameric rings of ClpP flanked by hexameric rings of ClpA. For full proteolytic activity ClpP must associate with one of two related ATPase subunits: ClpA and [[ClpX]]. |
</StructureSection> | </StructureSection> | ||
==3D structures of Clp protease== | ==3D structures of Clp protease== | ||
Revision as of 09:35, 31 January 2017
| |||||||||||
3D structures of Clp protease
Updated on 31-January-2017