Nuclear polyadenylated RNA-binding protein
From Proteopedia
| Line 1: | Line 1: | ||
=Introduction= | =Introduction= | ||
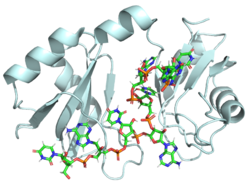
[[Image:Hrp1 cropped1.png|250 px|right|thumb|Figure 1: Cartoon representation of the Hrp1-PEE complex. The RNA is shown as a stick model and is colored by element.]] | [[Image:Hrp1 cropped1.png|250 px|right|thumb|Figure 1: Cartoon representation of the Hrp1-PEE complex. The RNA is shown as a stick model and is colored by element.]] | ||
| - | Hrp1 is a [https://en.wikipedia.org/wiki/Polyadenylation polyadenylation] factor found in Saccharomyces cervisiae (yeast) <ref name="GM3H">PMID: 16794580</ref>. This protein recognizes and binds to an RNA sequence in the [https://en.wikipedia.org/wiki/Three_prime_untranslated_region 3'UTR] of the [https://en.wikipedia.org/wiki/Messenger_RNA messenger RNA (mRNA)] upstream from the cleavage site called the polyadenylation enhancement element (PEE) <ref name="GM3H"/>. Upon binding to the RNA, Hrp1 helps recruit additional proteins necessary for the cleavage and polyadenylation of the RNA molecule <ref name="GM3H"/>. Although Hrp1 shares several common features with other RNA-binding proteins, the unique structural features of the Hrp1-PEE complex reveals the mechanism by which Hrp1 is able to recognize and bind to its specific RNA sequence at the atomic level <ref name="GM3H"/>. | + | Hrp1 is a [https://en.wikipedia.org/wiki/Polyadenylation polyadenylation] factor found in Saccharomyces cervisiae (yeast) <ref name="GM3H">PMID: 16794580</ref>. It is part of the Cleavage Factor I (CFI) B component. As mentioned below, Hrp1 of CFIB interacts with Rna14 and Rna15 of CFIA. These complexes aid in cleavage, polyadenylation, and transport of the mRNA from the nucleus. This protein recognizes and binds to an RNA sequence in the [https://en.wikipedia.org/wiki/Three_prime_untranslated_region 3'UTR] of the [https://en.wikipedia.org/wiki/Messenger_RNA messenger RNA (mRNA)] upstream from the cleavage site called the polyadenylation enhancement element (PEE) <ref name="GM3H"/>. Upon binding to the RNA, Hrp1 helps recruit additional proteins necessary for the cleavage and polyadenylation of the RNA molecule <ref name="GM3H"/>. Although Hrp1 shares several common features with other RNA-binding proteins, the unique structural features of the Hrp1-PEE complex reveals the mechanism by which Hrp1 is able to recognize and bind to its specific RNA sequence at the atomic level <ref name="GM3H"/>. |
Revision as of 15:52, 3 April 2018
Contents |
Introduction
Hrp1 is a polyadenylation factor found in Saccharomyces cervisiae (yeast) [1]. It is part of the Cleavage Factor I (CFI) B component. As mentioned below, Hrp1 of CFIB interacts with Rna14 and Rna15 of CFIA. These complexes aid in cleavage, polyadenylation, and transport of the mRNA from the nucleus. This protein recognizes and binds to an RNA sequence in the 3'UTR of the messenger RNA (mRNA) upstream from the cleavage site called the polyadenylation enhancement element (PEE) [1]. Upon binding to the RNA, Hrp1 helps recruit additional proteins necessary for the cleavage and polyadenylation of the RNA molecule [1]. Although Hrp1 shares several common features with other RNA-binding proteins, the unique structural features of the Hrp1-PEE complex reveals the mechanism by which Hrp1 is able to recognize and bind to its specific RNA sequence at the atomic level [1].
Structure
| |||||||||||
Interaction with RNA15
RNA15 is another RNA-binding protein with a single N-terminal RNA recognition motif (RRM) [2]. RNA15 recognizes an A-rich positioning element (PE) downstream from the PEE but upstream from the 3' cleavage site [2]. The recognition of the PE by RNA15 is crucial for precise cleavage of the RNA molecule. Hrp1 and RNA15 are held together by a separate protein, RNA14 [2]. These proteins act together to anchor the polyadenylation and cleavage protein machinery relative to the cleavage site for precise 3'-end processing [2].
Relationship to other proteins
The RNP-type RBD is found in many proteins involved in post-transcriptional pre-mRNA processing (5'-end capping, splicing, 3'-end cleavage and polyadenylation, and transport from the nucleus)[3]. The unique RBD of Hrp1 enables the protein to bind an RNA sequence that differs in both length and content from the RNA sequences of other RNA-binding and mRNA processing proteins such as sex lethal, Poly (A)-binding protein (PABP), and HuD [1]. Like Hrp1, each of these proteins belong to the class of single strand proteins composed of two canonical RBDs; however, these proteins are differentiated by their target RNA sequence, their interactions with RNA at the atomic level, and their interdomain contacts [1]. One way in which Hrp1 differentiates itself from these other proteins is by the fact that Hud, sex lethal, and PABP all contain at least one intra-RNA base-base stacking interaction, a feature that is not found in the Hrp1-PEE complex [1]. It is possible that the intra-RNA interactions found in these other proteins is replaced by the crucial Trp168-Ade4 stacking interaction found in the Hrp1 complex [1]. The fact that the intra-RNA base-base stacking interactions are replaced by the Trp168-Ade4 in the Hrp1-PEE complex might also explain why the Hrp1-RNA interface involves only 6 nucleotides whereas PABP, sex lethal, and HuD require a longer 8-10 nucleotide sequence in the RNA binding pocket [1].
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 1.10 1.11 1.12 1.13 1.14 1.15 1.16 1.17 1.18 1.19 1.20 1.21 Perez-Canadillas JM. Grabbing the message: structural basis of mRNA 3'UTR recognition by Hrp1. EMBO J. 2006 Jul 12;25(13):3167-78. Epub 2006 Jun 22. PMID:16794580
- ↑ 2.0 2.1 2.2 2.3 Leeper TC, Qu X, Lu C, Moore C, Varani G. Novel protein-protein contacts facilitate mRNA 3'-processing signal recognition by Rna15 and Hrp1. J Mol Biol. 2010 Aug 20;401(3):334-49. Epub 2010 Jun 19. PMID:20600122 doi:10.1016/j.jmb.2010.06.032
- ↑ Clery A, Blatter M, Allain FH. RNA recognition motifs: boring? Not quite. Curr Opin Struct Biol. 2008 Jun;18(3):290-8. doi: 10.1016/j.sbi.2008.04.002. PMID:18515081 doi:http://dx.doi.org/10.1016/j.sbi.2008.04.002
Proteopedia Page Contributors and Editors (what is this?)
Cory A. Wuerch, Matthew Douglas Moore, Savannah Davis, Michal Harel, Jaime Prilusky