This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Andrea Foote/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 5: | Line 5: | ||
== Structure == | == Structure == | ||
| - | Purα functions as a dimer composed of two intramolecular domains and one intermolecular domain. The Purα monomer contains three semi-conserved repeated amino acid sequences, named in order from N->C: PUR repeats I, II, and III. These repeats fold to form two domains: <scene name='78/786627/5fgp_intro/8'>PUR repeats I and II</scene> associating to form the I-II domain or “intramolecular domain”, while <scene name='78/786627/5fgo_repeatiii/2'>PUR repeat III</scene> facilitates dimerization through association with a repeat III from a second Purα monomer or repeat III of Purβ. Each PUR repeat is connected by flexible linker regions. | ||
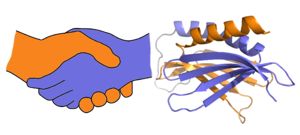
[[Image:180429 proteopedia pura figures2.jpg|thumb|right|300px| A PUR domain is analogous to a left-handed handshake. PUR repeat I-II represented from 5fgp.]] | [[Image:180429 proteopedia pura figures2.jpg|thumb|right|300px| A PUR domain is analogous to a left-handed handshake. PUR repeat I-II represented from 5fgp.]] | ||
| + | Purα is a Purα functions as a dimer composed of two intramolecular domains and one intermolecular domain. The Purα monomer contains three semi-conserved repeated amino acid sequences, named in order from N->C: PUR repeats I, II, and III. These repeats fold to form two domains: <scene name='78/786627/5fgp_intro/8'>PUR repeats I and II</scene> associating to form the I-II domain or “intramolecular domain”, while <scene name='78/786627/5fgo_repeatiii/2'>PUR repeat III</scene> facilitates dimerization through association with a repeat III from a second Purα monomer or repeat III of Purβ. Each PUR repeat is connected by flexible linker regions. Each PUR repeat contains a beta-sheet composed of four beta-strands, followed by a single alpha-helix. | ||
| + | |||
| + | The domains of Purα have been described as "Whirly-like" folds because of their structural similarity to the DNA-binding Whirly class of proteins found in plants.<ref>PMID:19846792</ref> | ||
== Function == | == Function == | ||
High-affinity nucleic acid-binding function is dependent on Purα dimerization. Purα forms homodimers in addition to heterodimers with Purβ. PurA is known to repress various genes including | High-affinity nucleic acid-binding function is dependent on Purα dimerization. Purα forms homodimers in addition to heterodimers with Purβ. PurA is known to repress various genes including | ||
Revision as of 10:59, 3 May 2018
Purine-rich element binding protein alpha
| |||||||||||