This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
LDL receptor
From Proteopedia
(Difference between revisions)
| Line 12: | Line 12: | ||
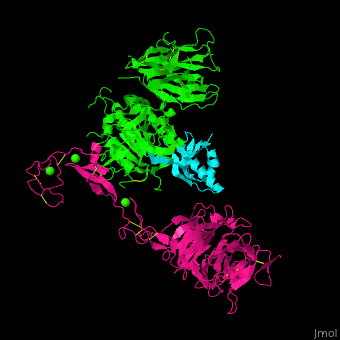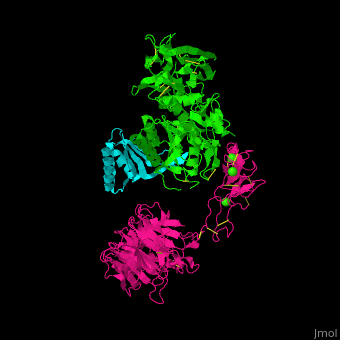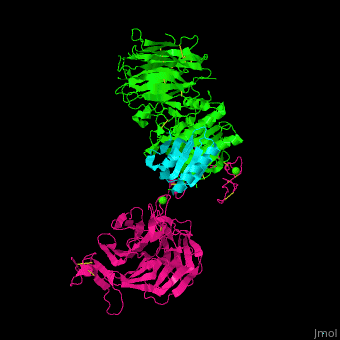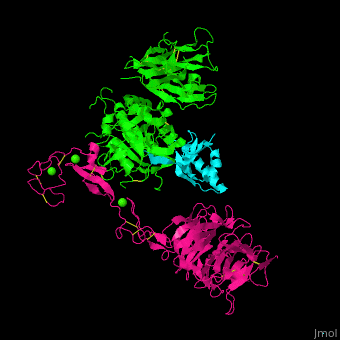
<scene name='54/545853/Cv/5'>Ligand-binding domain (LBD) and epidermal growth factor precursor homology domain (EGFP) of LDLR</scene>. | <scene name='54/545853/Cv/5'>Ligand-binding domain (LBD) and epidermal growth factor precursor homology domain (EGFP) of LDLR</scene>. | ||
| - | LDLR consists of a ligand-binding domain (LBD residues 1-292), epidermal growth factor precursor homology domain (EGFP residues 293-699), oligosaccharide-rich domain (residues 700-758), membrane-spanning domain (residues 759-781) and cytoplasmic domain (residues 782-832). LDLR LBD contains 7 ca. 40 amino acid long repeats ( | + | LDLR consists of a ligand-binding domain (LBD residues 1-292), epidermal growth factor precursor homology domain (EGFP residues 293-699), oligosaccharide-rich domain (residues 700-758), membrane-spanning domain (residues 759-781) and cytoplasmic domain (residues 782-832). LDLR LBD contains 7 ca. 40 amino acid long repeats ((LB1 residues 20-67; LB2 residues 55-104; LB3 residues 105-143; LB4 residues 144-196; LB5 residues 196-232; LB6 residues 234-272) containing 6 cysteine residues, making a calcium binding octahedral structure. LDLR EGFP contains 2 EGF repeats followed by 6 <scene name='54/545853/Cv/2'>YWTD repeats</scene> and another EGF repeat. LDLR LBD residues 133-273 are named C-type lectin-like domain. |
</StructureSection> | </StructureSection> | ||
| Line 35: | Line 35: | ||
**[[2fcw]] – hLDLR LB3,LB4 + α-2-macroglobulin receptor-associated protein<br /> | **[[2fcw]] – hLDLR LB3,LB4 + α-2-macroglobulin receptor-associated protein<br /> | ||
**[[2kri]] – hLDLR LB4 + β-2-glycoprotein - NMR<br /> | **[[2kri]] – hLDLR LB4 + β-2-glycoprotein - NMR<br /> | ||
| + | **[[5oyl]] – hLDLR LBD LB2 + glycoprotein <br /> | ||
| + | **[[5oy9]] – hLDLR LBD LB3 + glycoprotein G<br /> | ||
*hLDLR EGFP | *hLDLR EGFP | ||
Revision as of 09:19, 30 July 2018
| |||||||||||
3D structures of LDL receptor
Updated on 30-July-2018
References
- ↑ Goldstein JL, Brown MS. The LDL receptor. Arterioscler Thromb Vasc Biol. 2009 Apr;29(4):431-8. doi:, 10.1161/ATVBAHA.108.179564. PMID:19299327 doi:http://dx.doi.org/10.1161/ATVBAHA.108.179564
- ↑ Mak YT, Pang CP, Tomlinson B, Zhang J, Chan YS, Mak TW, Masarei JR. Mutations in the low-density lipoprotein receptor gene in Chinese familial hypercholesterolemia patients. Arterioscler Thromb Vasc Biol. 1998 Oct;18(10):1600-5. PMID:9763532