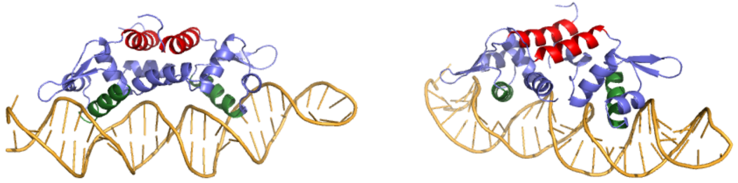This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
CzrA
From Proteopedia
(Difference between revisions)
(New page: =Zinc Dependent Transcriptional Repressor of the Czr operon (CzrA)= <StructureSection load='CzrAwithDNA.pdb' size='340' frame='true' side='right' caption='The dimer Czr A' scene=''> <scene...) |
|||
| (2 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | =Zinc Dependent Transcriptional Repressor of the Czr operon (CzrA)= | ||
<StructureSection load='CzrAwithDNA.pdb' size='340' frame='true' side='right' caption='The dimer Czr A' scene=''> | <StructureSection load='CzrAwithDNA.pdb' size='340' frame='true' side='right' caption='The dimer Czr A' scene=''> | ||
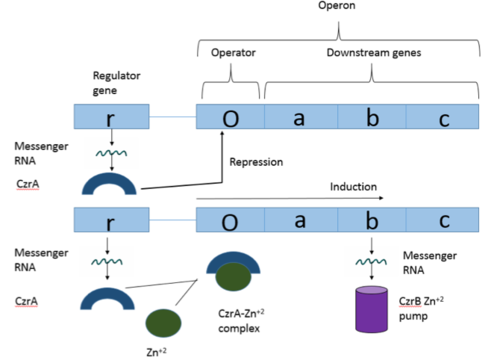
<scene name='69/694220/Czra_with_dna/1'>CzrA</scene> is a transcriptional repressor protein responsible for the regulation of the Czr operon in prokaryotes. The best studied example to date comes from ''Staphylococcus aureus''. | <scene name='69/694220/Czra_with_dna/1'>CzrA</scene> is a transcriptional repressor protein responsible for the regulation of the Czr operon in prokaryotes. The best studied example to date comes from ''Staphylococcus aureus''. | ||
| Line 28: | Line 27: | ||
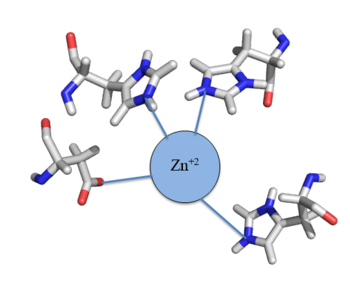
Zn<sup>+2</sup> binding is driven by a large [https://en.wikipedia.org/wiki/Entropy entropic] gain <ref>DOI:10.1021/ja906131b</ref>. Water molecules around the metal ion and Czr A protein are displaced, and gain greater freedom. This gain in entropy allows Zn<sup>+2</sup> to bind to Czr A with reasonable affinity and speed in vivo. The zinc<sup>+2</sup> ion forms a tetrahedral complex with the four residues (Figure 4). Other metal ions that may form a tetrahedral complex will have some affinity for Czr A; however, the metal binding pocket of Czr A has been optimized to bind Zn<sup>+2</sup> with the highest affinity. | Zn<sup>+2</sup> binding is driven by a large [https://en.wikipedia.org/wiki/Entropy entropic] gain <ref>DOI:10.1021/ja906131b</ref>. Water molecules around the metal ion and Czr A protein are displaced, and gain greater freedom. This gain in entropy allows Zn<sup>+2</sup> to bind to Czr A with reasonable affinity and speed in vivo. The zinc<sup>+2</sup> ion forms a tetrahedral complex with the four residues (Figure 4). Other metal ions that may form a tetrahedral complex will have some affinity for Czr A; however, the metal binding pocket of Czr A has been optimized to bind Zn<sup>+2</sup> with the highest affinity. | ||
| + | |||
| + | == 3D Structures of CzrA == | ||
| + | |||
| + | [[CzrA 3D structures]] | ||
</StructureSection> | </StructureSection> | ||
Current revision
| |||||||||||
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Arunkumar A., Campanello G., Giedroc D. (2009). Solution Structure of a paradigm ArsR family zinc sensor in the DNA-bound state. PNAS 106:43 18177-18182.
- ↑ Chakravorty DK, Wang B, Lee CW, Giedroc DP, Merz KM Jr. Simulations of allosteric motions in the zinc sensor CzrA. J Am Chem Soc. 2012 Feb 22;134(7):3367-76. doi: 10.1021/ja208047b. Epub 2011 Nov , 14. PMID:22007899 doi:http://dx.doi.org/10.1021/ja208047b
- ↑ MacPherson S, Larochelle M, Turcotte B. A fungal family of transcriptional regulators: the zinc cluster proteins. Microbiol Mol Biol Rev. 2006 Sep;70(3):583-604. PMID:16959962 doi:http://dx.doi.org/10.1128/MMBR.00015-06
- ↑ Miller J, McLachlan AD, Klug A. Repetitive zinc-binding domains in the protein transcription factor IIIA from Xenopus oocytes. EMBO J. 1985 Jun 4;4(6):1609-1614.
- ↑ Grossoehme NE, Giedroc DP. Energetics of allosteric negative coupling in the zinc sensor S. aureus CzrA. J Am Chem Soc. 2009 Dec 16;131(49):17860-70. doi: 10.1021/ja906131b. PMID:19995076 doi:http://dx.doi.org/10.1021/ja906131b
Student Contributors
- Katelyn Baumer
- Jakob Jozwiakowski
- Catie Liggett