This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Victor Reverte/Sandbox 1
From Proteopedia
(Difference between revisions)
| Line 12: | Line 12: | ||
Being assembled as a tetramer, MCU monomers posses 351 amino acid residues. Each subunit can be divided into four structural domains, them being N-terminal domain (NTD), linker helix domain (LHD), coiled-coil domain (CCD), and transmembrane domain (TMD). CCD and TMD are the pore-forming subunits, while LHD links this regions to NTD. Recently, regulation of the complex and dimerization of two tetrameres were reported as functions of NTD. | Being assembled as a tetramer, MCU monomers posses 351 amino acid residues. Each subunit can be divided into four structural domains, them being N-terminal domain (NTD), linker helix domain (LHD), coiled-coil domain (CCD), and transmembrane domain (TMD). CCD and TMD are the pore-forming subunits, while LHD links this regions to NTD. Recently, regulation of the complex and dimerization of two tetrameres were reported as functions of NTD. | ||
To guarantee selectivity, <scene name='81/817978/260wdimep265/1'>260WDIMEP265</scene>, a highly conserved sequence among protein homologues, from each monomer form two filters. The first one, dependent of <scene name='81/817978/D261/1'>D261 residues</scene> has a radius of affinity for hydrated calcium. The narrower one, is stabilized by <scene name='81/817978/E264/2'>E264</scene> and its selective for calcium radius. Finally, there’s a second constriction point at the end of the pore, formed by residues <scene name='81/817978/E288_and_v290/1'>E288 and V290</scene> of each monomer, that are involved in a juxtamembrane loop (JML). | To guarantee selectivity, <scene name='81/817978/260wdimep265/1'>260WDIMEP265</scene>, a highly conserved sequence among protein homologues, from each monomer form two filters. The first one, dependent of <scene name='81/817978/D261/1'>D261 residues</scene> has a radius of affinity for hydrated calcium. The narrower one, is stabilized by <scene name='81/817978/E264/2'>E264</scene> and its selective for calcium radius. Finally, there’s a second constriction point at the end of the pore, formed by residues <scene name='81/817978/E288_and_v290/1'>E288 and V290</scene> of each monomer, that are involved in a juxtamembrane loop (JML). | ||
| + | |||
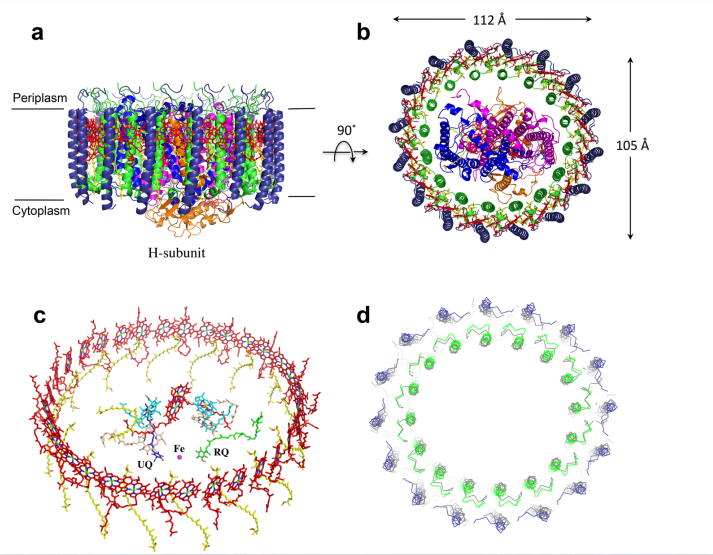
| + | [[Image:Structure.png]] | ||
| + | This image is from the article "Structural Mechanism of EMRE-Dependent Gating of the Human Mitochondrial Calcium Uniporter" by Youxing Jiang | ||
== Disease == | == Disease == | ||
Revision as of 17:02, 9 June 2019
Mitochondrial Calcium Uniporter (MCU)
| |||||||||||