This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
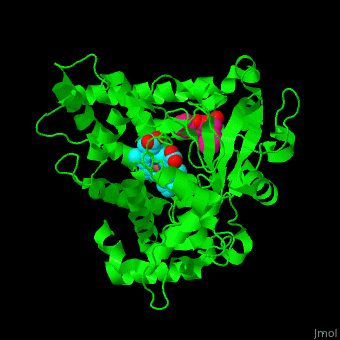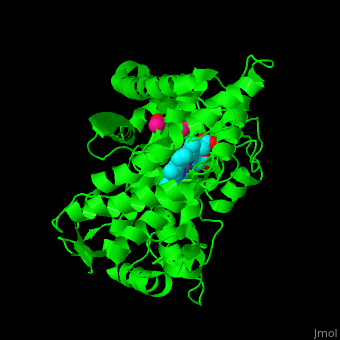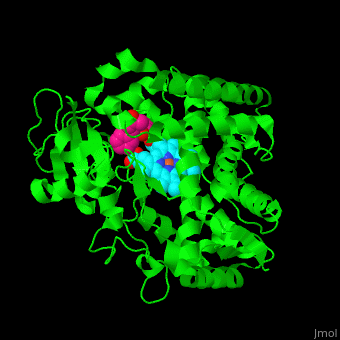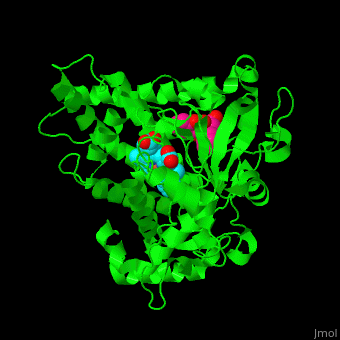
Cytochrome P450
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
== Function == | == Function == | ||
| - | [[Cytochrome P450]] (P450) catalyzes the oxidation of organic substances like lipids. The P450 contains a heme cofactor. The protein is numbered by its gene.<ref>PMID:12369887</ref>.<br /> | + | [[Cytochrome P450]] (P450) or '''cytochrome P450 monooxygenase''' catalyzes the oxidation of organic substances like lipids. The P450 contains a heme cofactor. The protein is numbered by its gene.<ref>PMID:12369887</ref>.<br /> |
* '''P450cam''', also known as camphor 5-monooxygenase, catalyzes the hydroxylation of camphor.<br /> | * '''P450cam''', also known as camphor 5-monooxygenase, catalyzes the hydroxylation of camphor.<br /> | ||
* '''P450nor''' is a Nitric Oxide Reductase P450.<br /> | * '''P450nor''' is a Nitric Oxide Reductase P450.<br /> | ||
Revision as of 09:30, 22 June 2021
| |||||||||||
References
- ↑ Danielson PB. The cytochrome P450 superfamily: biochemistry, evolution and drug metabolism in humans. Curr Drug Metab. 2002 Dec;3(6):561-97. PMID:12369887
- ↑ Williams PA, Cosme J, Ward A, Angove HC, Matak Vinkovic D, Jhoti H. Crystal structure of human cytochrome P450 2C9 with bound warfarin. Nature. 2003 Jul 24;424(6947):464-8. Epub 2003 Jul 13. PMID:12861225 doi:http://dx.doi.org/10.1038/nature01862