This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1ybq
From Proteopedia
(Difference between revisions)
| Line 3: | Line 3: | ||
<StructureSection load='1ybq' size='340' side='right'caption='[[1ybq]], [[Resolution|resolution]] 2.00Å' scene=''> | <StructureSection load='1ybq' size='340' side='right'caption='[[1ybq]], [[Resolution|resolution]] 2.00Å' scene=''> | ||
== Structural highlights == | == Structural highlights == | ||
| - | <table><tr><td colspan='2'>[[1ybq]] is a 2 chain structure with sequence from [ | + | <table><tr><td colspan='2'>[[1ybq]] is a 2 chain structure with sequence from [https://en.wikipedia.org/wiki/"bacillus_coli"_migula_1895 "bacillus coli" migula 1895]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1YBQ OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=1YBQ FirstGlance]. <br> |
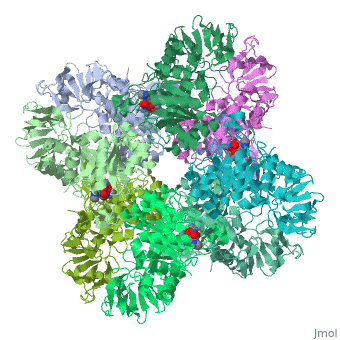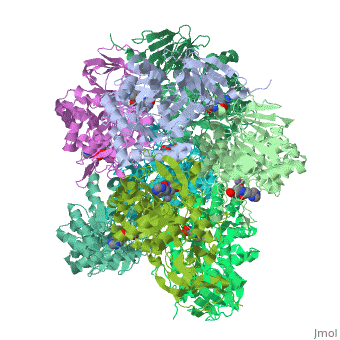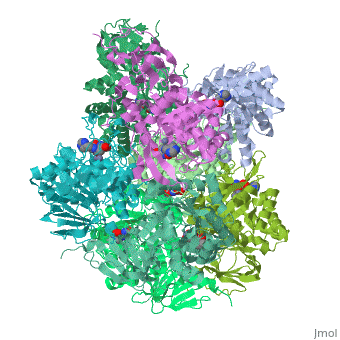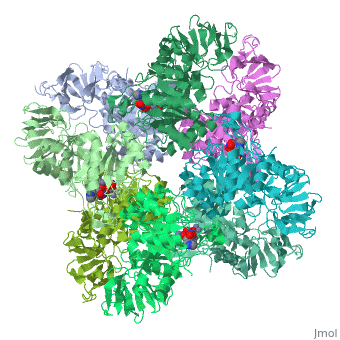
</td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=BDH:L-BETA-ASPARTYLHISTIDINE'>BDH</scene>, <scene name='pdbligand=ZN:ZINC+ION'>ZN</scene></td></tr> | </td></tr><tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=BDH:L-BETA-ASPARTYLHISTIDINE'>BDH</scene>, <scene name='pdbligand=ZN:ZINC+ION'>ZN</scene></td></tr> | ||
<tr id='NonStdRes'><td class="sblockLbl"><b>[[Non-Standard_Residue|NonStd Res:]]</b></td><td class="sblockDat"><scene name='pdbligand=KCX:LYSINE+NZ-CARBOXYLIC+ACID'>KCX</scene></td></tr> | <tr id='NonStdRes'><td class="sblockLbl"><b>[[Non-Standard_Residue|NonStd Res:]]</b></td><td class="sblockDat"><scene name='pdbligand=KCX:LYSINE+NZ-CARBOXYLIC+ACID'>KCX</scene></td></tr> | ||
| - | <tr id='gene'><td class="sblockLbl"><b>[[Gene|Gene:]]</b></td><td class="sblockDat">iadA ([ | + | <tr id='gene'><td class="sblockLbl"><b>[[Gene|Gene:]]</b></td><td class="sblockDat">iadA ([https://www.ncbi.nlm.nih.gov/Taxonomy/Browser/wwwtax.cgi?mode=Info&srchmode=5&id=562 "Bacillus coli" Migula 1895])</td></tr> |
| - | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=1ybq FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1ybq OCA], [https://pdbe.org/1ybq PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=1ybq RCSB], [https://www.ebi.ac.uk/pdbsum/1ybq PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=1ybq ProSAT]</span></td></tr> |
</table> | </table> | ||
== Function == | == Function == | ||
| - | [[ | + | [[https://www.uniprot.org/uniprot/IADA_ECOLI IADA_ECOLI]] Catalyzes the hydrolytic cleavage of a subset of L-isoaspartyl (L-beta-aspartyl) dipeptides. Used to degrade proteins damaged by L-isoaspartyl residues formation. The best substrate for the enzyme reported thus far is iso-Asp-Leu.<ref>PMID:7876157</ref> <ref>PMID:4880759</ref> <ref>PMID:12718528</ref> <ref>PMID:15882050</ref> |
== Evolutionary Conservation == | == Evolutionary Conservation == | ||
[[Image:Consurf_key_small.gif|200px|right]] | [[Image:Consurf_key_small.gif|200px|right]] | ||
Revision as of 16:50, 20 October 2021
Crystal structure of Escherichia coli isoaspartyl dipeptidase mutant D285N complexed with beta-aspartylhistidine
| |||||||||||