This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox Reserved 1732
From Proteopedia
(Difference between revisions)
| Line 22: | Line 22: | ||
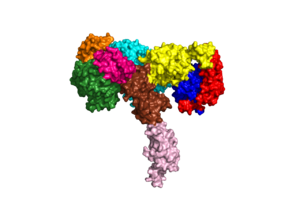
An insulin receptor is a dimer of heterodimers. The dimers are noncovalent, but the insulin receptors are covalently maintained as functional dimers by disulfide bonds. An insulin receptor is comprised of 2 α-chains, and 2 β-chains. The α-chain and an estimated 190 residues of the β-chain are located on the extracellular side of the plasma membrane. The rest of the beta-chain consists of a single transmembrane helix, the juxtamembrane domain, and the intracellular tyrosine kinase domain. | An insulin receptor is a dimer of heterodimers. The dimers are noncovalent, but the insulin receptors are covalently maintained as functional dimers by disulfide bonds. An insulin receptor is comprised of 2 α-chains, and 2 β-chains. The α-chain and an estimated 190 residues of the β-chain are located on the extracellular side of the plasma membrane. The rest of the beta-chain consists of a single transmembrane helix, the juxtamembrane domain, and the intracellular tyrosine kinase domain. | ||
The alpha subunits are the site for insulin binding. Each subunit is comprised of 2 Leucine rich domains (L1 and L2), a Cysteine rich domain (CR) and an α-chain C-terminal helix (α-CT). The two subunits are held together by a disulfide bond between the cysteine rich domains. | The alpha subunits are the site for insulin binding. Each subunit is comprised of 2 Leucine rich domains (L1 and L2), a Cysteine rich domain (CR) and an α-chain C-terminal helix (α-CT). The two subunits are held together by a disulfide bond between the cysteine rich domains. | ||
| + | |||
[[Image:6CE7.png]] | [[Image:6CE7.png]] | ||
| + | |||
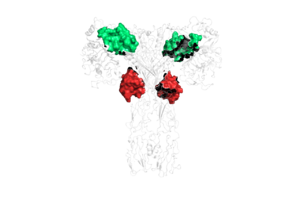
Due to the heterodimeric nature of the receptor, there are two types of insulin binding sites that are split into pairs in the alpha subunits: sites 1 and 1' and sites 2 and 2', for a total of 4 binding sites of insulin. Binding sites 1 and 1' have a greater surface area and are more easily accessible for the insulin to bond, resulting in a higher affinity for insulin binding. Binding sites 2 and 2' have less surface area and are located on the back of the beta sheet so their binding sites do not get filled as quickly. | Due to the heterodimeric nature of the receptor, there are two types of insulin binding sites that are split into pairs in the alpha subunits: sites 1 and 1' and sites 2 and 2', for a total of 4 binding sites of insulin. Binding sites 1 and 1' have a greater surface area and are more easily accessible for the insulin to bond, resulting in a higher affinity for insulin binding. Binding sites 2 and 2' have less surface area and are located on the back of the beta sheet so their binding sites do not get filled as quickly. | ||
| + | |||
[[Image:biochemIBSpt2.png]] | [[Image:biochemIBSpt2.png]] | ||
Revision as of 18:38, 14 November 2022
| This Sandbox is Reserved from August 30, 2022 through May 31, 2023 for use in the course Biochemistry I taught by Kimberly Lane at the Radford University, Radford, VA, USA. This reservation includes Sandbox Reserved 1730 through Sandbox Reserved 1749. |
To get started:
More help: Help:Editing |
Your Heading Here (maybe something like 'Structure')
| |||||||||||
References
1. PDB101: Molecule of the month: Insulin receptor https://pdb101.rcsb.org/motm/182 (accessed Oct 21, 2022).