This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1pvo
From Proteopedia
(Difference between revisions)
| (12 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | [[Image:1pvo.gif|left|200px]] | ||
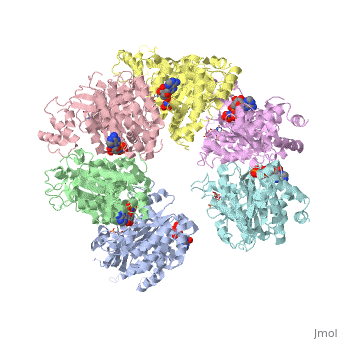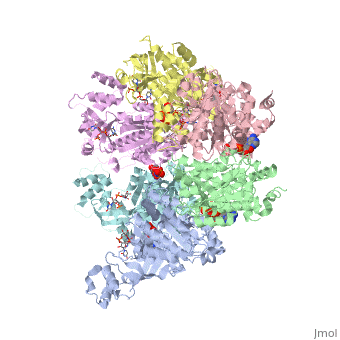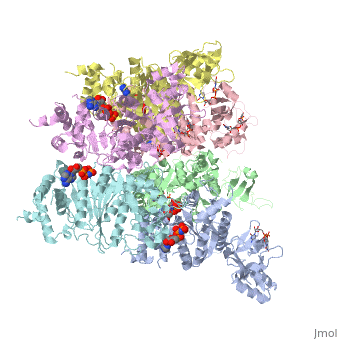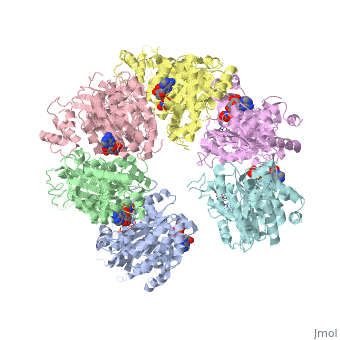
| - | < | + | ==X-ray crystal structure of Rho transcription termination factor in complex with ssRNA substrate and ANPPNP== |
| - | + | <StructureSection load='1pvo' size='340' side='right'caption='[[1pvo]], [[Resolution|resolution]] 3.00Å' scene=''> | |
| - | You may | + | == Structural highlights == |
| - | + | <table><tr><td colspan='2'>[[1pvo]] is a 11 chain structure with sequence from [https://en.wikipedia.org/wiki/Escherichia_coli Escherichia coli]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1PVO OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=1PVO FirstGlance]. <br> | |
| - | + | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 3Å</td></tr> | |
| - | - | + | <tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=ANP:PHOSPHOAMINOPHOSPHONIC+ACID-ADENYLATE+ESTER'>ANP</scene></td></tr> |
| - | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=1pvo FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1pvo OCA], [https://pdbe.org/1pvo PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=1pvo RCSB], [https://www.ebi.ac.uk/pdbsum/1pvo PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=1pvo ProSAT]</span></td></tr> | |
| + | </table> | ||
| + | == Function == | ||
| + | [https://www.uniprot.org/uniprot/RHO_ECOLI RHO_ECOLI] Facilitates transcription termination by a mechanism that involves rho binding to the nascent RNA, activation of rho's RNA-dependent ATPase activity, and release of the mRNA from the DNA template. RNA-dependent NTPAse which utilizes all four ribonucleoside triphosphates as substrates.[HAMAP-Rule:MF_01884] | ||
| + | == Evolutionary Conservation == | ||
| + | [[Image:Consurf_key_small.gif|200px|right]] | ||
| + | Check<jmol> | ||
| + | <jmolCheckbox> | ||
| + | <scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/pv/1pvo_consurf.spt"</scriptWhenChecked> | ||
| + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
| + | <text>to colour the structure by Evolutionary Conservation</text> | ||
| + | </jmolCheckbox> | ||
| + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=1pvo ConSurf]. | ||
| + | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
| + | In bacteria, one of the major transcriptional termination mechanisms requires a RNA/DNA helicase known as the Rho factor. We have determined two structures of Rho complexed with nucleic acid recognition site mimics in both free and nucleotide bound states to 3.0 A resolution. Both structures show that Rho forms a hexameric ring in which two RNA binding sites--a primary one responsible for target mRNA recognition and a secondary one required for mRNA translocation and unwinding--point toward the center of the ring. Rather than forming a closed ring, the Rho hexamer is split open, resembling a "lock washer" in its global architecture. The distance between subunits at the opening is sufficiently wide (12 A) to accommodate single-stranded RNA. This open configuration most likely resembles a state poised to load onto mRNA and suggests how related ring-shaped enzymes may be breached to bind nucleic acids. | ||
| - | + | Structure of the Rho transcription terminator: mechanism of mRNA recognition and helicase loading.,Skordalakes E, Berger JM Cell. 2003 Jul 11;114(1):135-46. PMID:12859904<ref>PMID:12859904</ref> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |
| - | + | </div> | |
| + | <div class="pdbe-citations 1pvo" style="background-color:#fffaf0;"></div> | ||
| - | == | + | ==See Also== |
| - | + | *[[Helicase 3D structures|Helicase 3D structures]] | |
| + | == References == | ||
| + | <references/> | ||
| + | __TOC__ | ||
| + | </StructureSection> | ||
[[Category: Escherichia coli]] | [[Category: Escherichia coli]] | ||
| - | [[Category: | + | [[Category: Large Structures]] |
| - | [[Category: Berger | + | [[Category: Berger JM]] |
| - | [[Category: Skordalakes | + | [[Category: Skordalakes E]] |
| - | + | ||
| - | + | ||
Current revision
X-ray crystal structure of Rho transcription termination factor in complex with ssRNA substrate and ANPPNP
| |||||||||||