This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
4iou
From Proteopedia
(Difference between revisions)
| (One intermediate revision not shown.) | |||
| Line 3: | Line 3: | ||
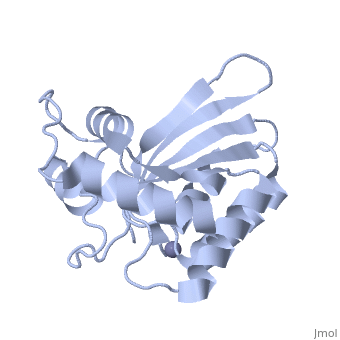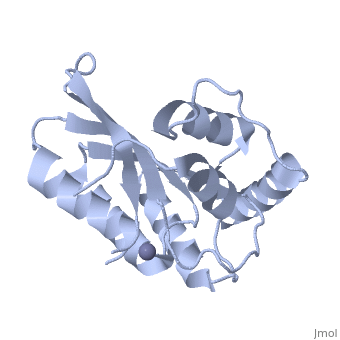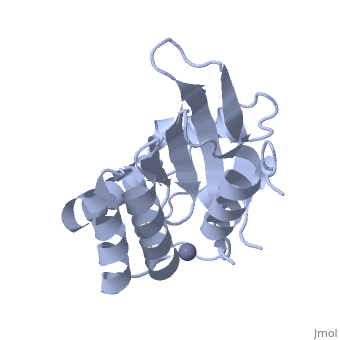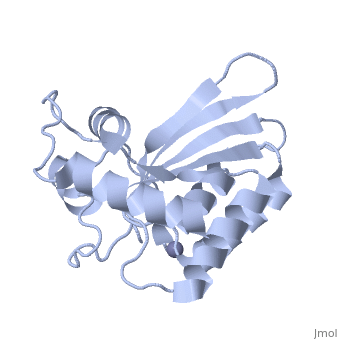
<StructureSection load='4iou' size='340' side='right'caption='[[4iou]], [[Resolution|resolution]] 2.75Å' scene=''> | <StructureSection load='4iou' size='340' side='right'caption='[[4iou]], [[Resolution|resolution]] 2.75Å' scene=''> | ||
== Structural highlights == | == Structural highlights == | ||
| - | <table><tr><td colspan='2'>[[4iou]] is a 4 chain structure with sequence from [ | + | <table><tr><td colspan='2'>[[4iou]] is a 4 chain structure with sequence from [https://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=4IOU OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=4IOU FirstGlance]. <br> |
| - | </td></tr><tr id=' | + | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 2.751Å</td></tr> |
| - | <tr id=' | + | <tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=ZN:ZINC+ION'>ZN</scene></td></tr> |
| - | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[ | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=4iou FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=4iou OCA], [https://pdbe.org/4iou PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=4iou RCSB], [https://www.ebi.ac.uk/pdbsum/4iou PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=4iou ProSAT]</span></td></tr> |
</table> | </table> | ||
== Function == | == Function == | ||
| - | [ | + | [https://www.uniprot.org/uniprot/ABC3F_HUMAN ABC3F_HUMAN] DNA deaminase (cytidine deaminase) which acts as an inhibitor of retrovirus replication and retrotransposon mobility via deaminase-dependent and -independent mechanisms. Exhibits antiviral activity against vif-deficient HIV-1. After the penetration of retroviral nucleocapsids into target cells of infection and the initiation of reverse transcription, it can induce the conversion of cytosine to uracil in the minus-sense single-strand viral DNA, leading to G-to-A hypermutations in the subsequent plus-strand viral DNA. The resultant detrimental levels of mutations in the proviral genome, along with a deamination-independent mechanism that works prior to the proviral integration, together exert efficient antiretroviral effects in infected target cells. Selectively targets single-stranded DNA and does not deaminate double-stranded DNA or single- or double-stranded RNA. Exhibits antiviral activity also against hepatitis B virus (HBV), equine infectious anemia virus (EIAV), xenotropic MuLV-related virus (XMRV) and simian foamy virus (SFV) and may inhibit the mobility of LTR and non-LTR retrotransposons. May also play a role in the epigenetic regulation of gene expression through the process of active DNA demethylation.<ref>PMID:15152192</ref> <ref>PMID:16527742</ref> <ref>PMID:16378963</ref> <ref>PMID:19458006</ref> <ref>PMID:20219927</ref> <ref>PMID:20335265</ref> <ref>PMID:20062055</ref> <ref>PMID:21496894</ref> <ref>PMID:21835787</ref> <ref>PMID:22915799</ref> <ref>PMID:22807680</ref> <ref>PMID:23097438</ref> <ref>PMID:23152537</ref> |
<div style="background-color:#fffaf0;"> | <div style="background-color:#fffaf0;"> | ||
== Publication Abstract from PubMed == | == Publication Abstract from PubMed == | ||
| Line 23: | Line 23: | ||
__TOC__ | __TOC__ | ||
</StructureSection> | </StructureSection> | ||
| - | [[Category: | + | [[Category: Homo sapiens]] |
[[Category: Large Structures]] | [[Category: Large Structures]] | ||
| - | [[Category: Bohn | + | [[Category: Bohn M]] |
| - | [[Category: Schiffer | + | [[Category: Schiffer CA]] |
| - | [[Category: Shandilya | + | [[Category: Shandilya SMD]] |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
Current revision
Crystal structure of the HIV-1 Vif binding, catalytically active domain of APOBEC3F
| |||||||||||