This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sequestosome
From Proteopedia
(Difference between revisions)
(New page: <StructureSection load='3ade' size='340' side='right' caption='Structure of mouse sequestosome complex with KEAP-1 (PDB code 3ade)' scene=''> == Function == '''Sequestosome''' (SQS)...) |
|||
| (10 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
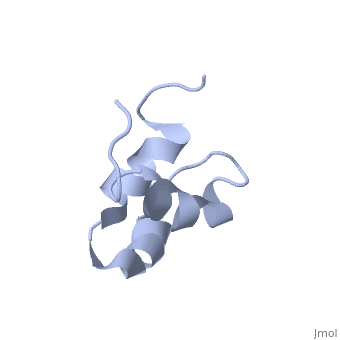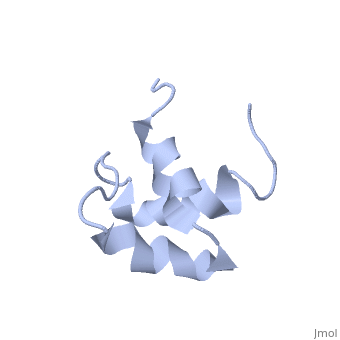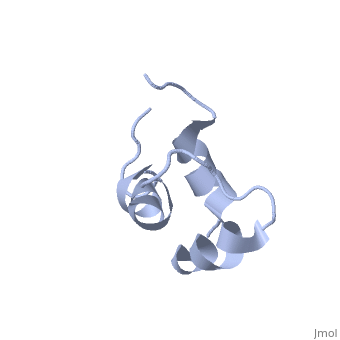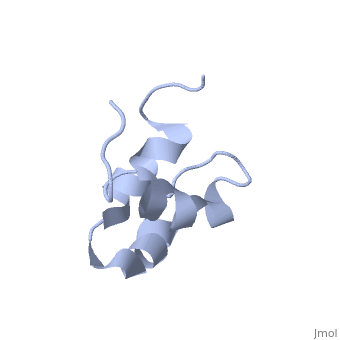
| - | + | <StructureSection load='2jy8' size='340' side='right' caption='Structure of human sequestosome UBA domain (PDB code [[2jy8]])' scene=''> | |
| - | <StructureSection load=' | + | |
== Function == | == Function == | ||
| + | '''Sequestosome''' (SQS) or '''p62''', is required for the formation and autophagic degredation of polyubiquitin-containg bodies. SQS may regulate signaling cascades through ubiquitination<ref>PMID:15340068</ref>. | ||
| - | + | == Structural highlights == | |
SQS is a multi-domain protein. SQS domain structure includes: <br /> | SQS is a multi-domain protein. SQS domain structure includes: <br /> | ||
| - | PB1 domain which contains Phox and Berm1<br /> | + | *PB1 domain which contains Phox and Berm1<br /> |
| - | ZZ-type zinc finger <br /> | + | *ZZ-type zinc finger <br /> |
| - | SMIR – SOD interaction domain<br /> | + | *SMIR – SOD interaction domain<br /> |
| - | TB – TRAF6 binding motif<br /> | + | *TB – TRAF6 binding motif<br /> |
| - | LC3 – microtubule-associated protein 1 light chain 3B<br /> | + | *LC3 – microtubule-associated protein 1 light chain 3B<br /> |
| - | LIR – interaction region<br /> | + | *LIR – interaction region<br /> |
| - | UBA – ubiquitin association domain. | + | *UBA – ubiquitin association domain. |
| - | + | ||
| - | + | ||
== Relevance == | == Relevance == | ||
| - | + | SQS is associated with fibrillary tangles in Alzheimer disease<ref>PMID:22138392</ref>. | |
| - | + | ||
==3D structures of sequestosome== | ==3D structures of sequestosome== | ||
| Line 24: | Line 21: | ||
Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | ||
| - | [[1q02]], [[2jy7]], [[2jy8]], [[2k0b]], [[2knv]] – | + | Domains: PB1 1-102; ZZ 120-180; UBA 387-436 |
| - | [[ | + | |
| - | [[ | + | [[1q02]], [[2jy7]], [[2jy8]], [[2k0b]], [[2knv]] – hSQS UBA domain – human - NMR<br /> |
| + | [[6jm4]] – hSQS-1 PB1 domain (mutant)<br /> | ||
| + | [[4uf8]], [[4uf9]], [[6tgy]], [[6th3]] – hSQS-1 PB1 domain – Cryo EM<br /> | ||
| + | [[5yp7]], [[5yp8]], [[5ypa]], [[5ypb]], [[5ypc]], [[5ype]], [[5ypg]], [[5yph]], [[5ypi]], [[6miu]] – hSQS-1 ZZ domain<br /> | ||
| + | [[6mj7]] – hSQS-1 ZZ domain + Arg<br /> | ||
| + | [[6khz]] – hSQS-1 ZZ domain + Gly<br /> | ||
| + | [[7r1o]] – hSQS-1 ZZ domain + drug<br /> | ||
[[2ktr]] – rSQS PB1 domain – rat - NMR<br /> | [[2ktr]] – rSQS PB1 domain – rat - NMR<br /> | ||
[[2kkc]] – rSQS PB1 domain (mutant) - NMR<br /> | [[2kkc]] – rSQS PB1 domain (mutant) - NMR<br /> | ||
| - | [[ | + | [[4mjs]] – hSQS-1 PB1 domain + protein kinase C zeta type<br /> |
| + | [[3b0f]] – mSQS UBA domain – mouse <br /> | ||
| + | [[2rru]] – mSQS UBA domain - NMR<br /> | ||
Current revision
| |||||||||||
References
- ↑ Seibenhener ML, Babu JR, Geetha T, Wong HC, Krishna NR, Wooten MW. Sequestosome 1/p62 is a polyubiquitin chain binding protein involved in ubiquitin proteasome degradation. Mol Cell Biol. 2004 Sep;24(18):8055-68. PMID:15340068 doi:10.1128/MCB.24.18.8055-8068.2004
- ↑ Salminen A, Kaarniranta K, Haapasalo A, Hiltunen M, Soininen H, Alafuzoff I. Emerging role of p62/sequestosome-1 in the pathogenesis of Alzheimer's disease. Prog Neurobiol. 2012 Jan;96(1):87-95. doi: 10.1016/j.pneurobio.2011.11.005. Epub , 2011 Nov 22. PMID:22138392 doi:http://dx.doi.org/10.1016/j.pneurobio.2011.11.005