This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
1v3v
From Proteopedia
(Difference between revisions)
(New page: 200px<br /><applet load="1v3v" size="450" color="white" frame="true" align="right" spinBox="true" caption="1v3v, resolution 2.0Å" /> '''Crystal structure of ...) |
|||
| (17 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | [[Image:1v3v.gif|left|200px]]<br /><applet load="1v3v" size="450" color="white" frame="true" align="right" spinBox="true" | ||
| - | caption="1v3v, resolution 2.0Å" /> | ||
| - | '''Crystal structure of leukotriene B4 12-hydroxydehydrogenase/15-oxo-prostaglandin 13-reductase complexed with NADP and 15-oxo-PGE2'''<br /> | ||
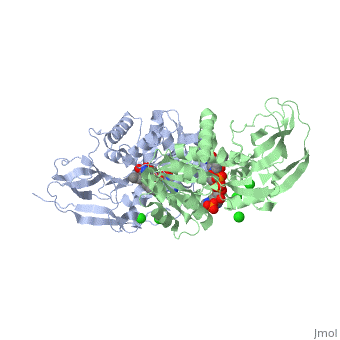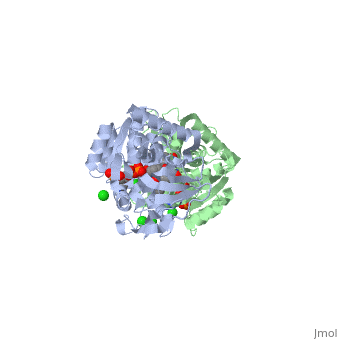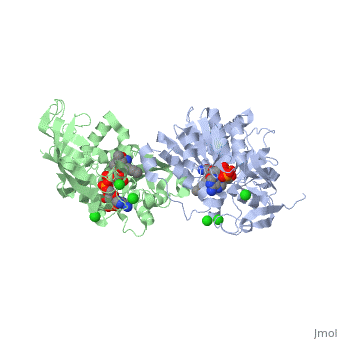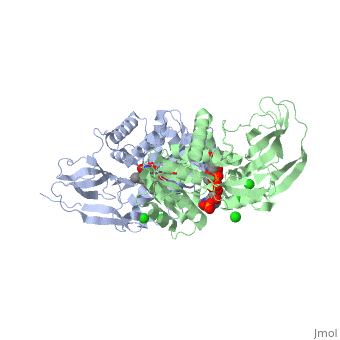
| - | == | + | ==Crystal structure of leukotriene B4 12-hydroxydehydrogenase/15-oxo-prostaglandin 13-reductase complexed with NADP and 15-oxo-PGE2== |
| - | The bifunctional leukotriene B(4) | + | <StructureSection load='1v3v' size='340' side='right'caption='[[1v3v]], [[Resolution|resolution]] 2.00Å' scene=''> |
| + | == Structural highlights == | ||
| + | <table><tr><td colspan='2'>[[1v3v]] is a 2 chain structure with sequence from [https://en.wikipedia.org/wiki/Cavia_porcellus Cavia porcellus]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1V3V OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=1V3V FirstGlance]. <br> | ||
| + | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 2Å</td></tr> | ||
| + | <tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=5OP:(5E,13E)-11-HYDROXY-9,15-DIOXOPROSTA-5,13-DIEN-1-OIC+ACID'>5OP</scene>, <scene name='pdbligand=CL:CHLORIDE+ION'>CL</scene>, <scene name='pdbligand=NAP:NADP+NICOTINAMIDE-ADENINE-DINUCLEOTIDE+PHOSPHATE'>NAP</scene></td></tr> | ||
| + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=1v3v FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1v3v OCA], [https://pdbe.org/1v3v PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=1v3v RCSB], [https://www.ebi.ac.uk/pdbsum/1v3v PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=1v3v ProSAT], [https://www.topsan.org/Proteins/RSGI/1v3v TOPSAN]</span></td></tr> | ||
| + | </table> | ||
| + | == Function == | ||
| + | [https://www.uniprot.org/uniprot/PTGR1_CAVPO PTGR1_CAVPO] Functions as 15-oxo-prostaglandin 13-reductase and acts on 15-oxo-PGE1, 15-oxo-PGE2 and 15-oxo-PGE2-alpha. Catalyzes the conversion of leukotriene B4 into its biologically less active metabolite, 12-oxo-leukotriene B4. This is an initial and key step of metabolic inactivation of leukotriene B4. | ||
| + | == Evolutionary Conservation == | ||
| + | [[Image:Consurf_key_small.gif|200px|right]] | ||
| + | Check<jmol> | ||
| + | <jmolCheckbox> | ||
| + | <scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/v3/1v3v_consurf.spt"</scriptWhenChecked> | ||
| + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
| + | <text>to colour the structure by Evolutionary Conservation</text> | ||
| + | </jmolCheckbox> | ||
| + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=1v3v ConSurf]. | ||
| + | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
| + | The bifunctional leukotriene B(4) 12-hydroxydehydrogenase/15-oxo-prostaglandin 13-reductase (LTB(4) 12-HD/PGR) is an essential enzyme for eicosanoid inactivation. It is involved in the metabolism of the E and F series of 15-oxo-prostaglandins (15-oxo-PGs), leukotriene B(4) (LTB(4)), and 15-oxo-lipoxin A(4) (15-oxo-LXA(4)). Some nonsteroidal anti-inflammatory drugs (NSAIDs), which primarily act as cyclooxygenase inhibitors also inhibit LTB(4) 12-HD/PGR activity. Here we report the crystal structure of the LTB(4) 12-HD/PGR, the binary complex structure with NADP(+), and the ternary complex structure with NADP(+) and 15-oxo-PGE(2). In the ternary complex, both in the crystalline form and in solution, the enolate anion intermediate accumulates as a brown chromophore. PGE(2) contains two chains, but only the omega-chain of 15-oxo-PGE(2) was defined in the electron density map in the ternary complex structure. The omega-chain was identified at the hydrophobic pore on the dimer interface. The structure showed that the 15-oxo group forms hydrogen bonds with the 2'-hydroxyl group of nicotine amide ribose of NADP(+) and a bound water molecule to stabilize the enolate intermediate during the reductase reaction. The electron-deficient C13 atom of the conjugated enolate may be directly attacked by a hydride from the NADPH nicotine amide in a stereospecific manner. The moderate recognition of 15-oxo-PGE(2) is consistent with a broad substrate specificity of LTB(4) 12-HD/PGR. The structure also implies that a Src homology domain 3 may interact with the left-handed proline-rich helix at the dimer interface and regulate LTB(4) 12-HD/PGR activity by disruption of the substrate binding pore to accommodate the omega-chain. | ||
| - | + | Structural basis of leukotriene B4 12-hydroxydehydrogenase/15-Oxo-prostaglandin 13-reductase catalytic mechanism and a possible Src homology 3 domain binding loop.,Hori T, Yokomizo T, Ago H, Sugahara M, Ueno G, Yamamoto M, Kumasaka T, Shimizu T, Miyano M J Biol Chem. 2004 May 21;279(21):22615-23. Epub 2004 Mar 8. PMID:15007077<ref>PMID:15007077</ref> | |
| - | + | ||
| - | + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |
| - | + | </div> | |
| - | + | <div class="pdbe-citations 1v3v" style="background-color:#fffaf0;"></div> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ==See Also== | |
| + | *[[Leukotriene B4 hydroxydehydrogenase|Leukotriene B4 hydroxydehydrogenase]] | ||
| + | == References == | ||
| + | <references/> | ||
| + | __TOC__ | ||
| + | </StructureSection> | ||
| + | [[Category: Cavia porcellus]] | ||
| + | [[Category: Large Structures]] | ||
| + | [[Category: Ago H]] | ||
| + | [[Category: Hori T]] | ||
| + | [[Category: Kumasaka T]] | ||
| + | [[Category: Miyano M]] | ||
| + | [[Category: Shimizu T]] | ||
| + | [[Category: Sugahara M]] | ||
| + | [[Category: Ueno G]] | ||
| + | [[Category: Yamamoto M]] | ||
| + | [[Category: Yokomizo T]] | ||
Current revision
Crystal structure of leukotriene B4 12-hydroxydehydrogenase/15-oxo-prostaglandin 13-reductase complexed with NADP and 15-oxo-PGE2
| |||||||||||
Categories: Cavia porcellus | Large Structures | Ago H | Hori T | Kumasaka T | Miyano M | Shimizu T | Sugahara M | Ueno G | Yamamoto M | Yokomizo T