This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Vinculin
From Proteopedia
(Difference between revisions)
| (10 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | |||
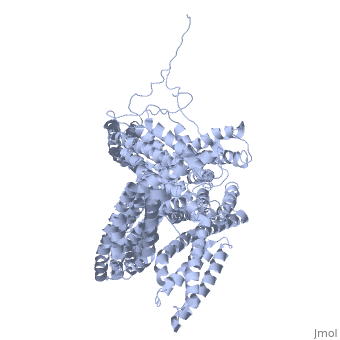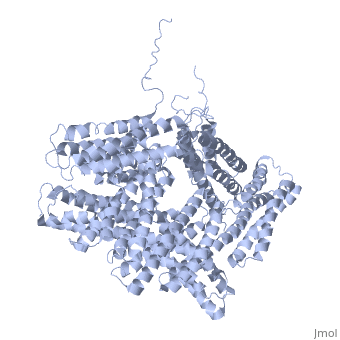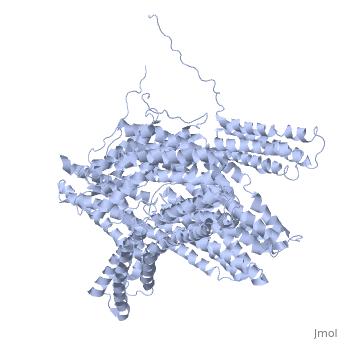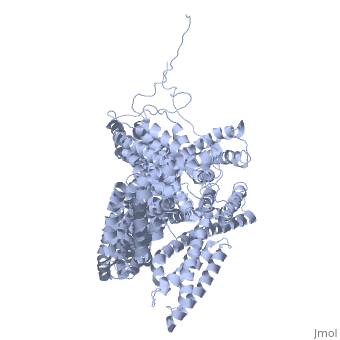
<StructureSection load='1st6' size='340' side='right' caption='Chicken full-length metavinculin, [[1st6]]' scene='' > | <StructureSection load='1st6' size='340' side='right' caption='Chicken full-length metavinculin, [[1st6]]' scene='' > | ||
| - | [[Vinculin|Vinculins]] (VCLs) are involved in adhesion by linking integrin molecules to the actin cytoskeleton. Its head domain (Vd1) can bind to [[Talin|talin]] or to [[Actinin|alpha-actinin]] at their respective VCL Binding Sites (VBS). | + | == Function == |
| - | + | [[Vinculin|Vinculins]] (VCLs) are involved in adhesion by linking integrin molecules to the actin cytoskeleton. Its head domain (Vd1) can bind to [[Talin|talin]] or to [[Actinin|alpha-actinin]] at their respective VCL Binding Sites (VBS)<ref>PMID:11152287</ref>. The protein raver1 RNA Recognition Motif (RRM) forms a complex with VCL or m-VCL. '''Metavinculin''' (m-VCL) is a splice version of VCL containing an extra ca. 70 amino acids in the C-terminal domain. | |
| - | + | ||
| - | + | == Relevance == | |
| - | + | Loss of VCL could be used as a prognostic factor for colorectal cancer se it promotes metastasis<ref>PMID:25496021</ref>. | |
| - | + | == Disease == | |
| + | Mutation in m-VCL can yield cardiomyopathic phenotype<ref>PMID:16236538</ref>. | ||
| - | + | == Structural highlights == | |
| - | + | <scene name='Sandbox_27/Role_i997_vinculin_head-tail_1/1'>Vinculin Autoinhibition</scene> is achieved through a high affinity intramolecular interaction between tail (orange) and head (aqua) domains ([[1st6]]). Energetically, I997 is key to maintaining this autoinhibition. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | == 3D Structures of Vinculin == | |
| + | [[Vinculin 3D structures]] | ||
| - | **[[3myi]] – m-VCL tail domain <br /> | ||
| - | **[[3rf3]] - hm-VCL + invasin IPAA<br /> | ||
| - | **[[3s90]] - hm-VCL head domain + mTalin-1 peptide<br /> | ||
| - | **[[4dj9]] - hm-VCL head domain + hTalin-1 peptide <br /> | ||
| - | **[[3tj5]] - hm-VCL head domain + Sca-family protein peptide<br /> | ||
| - | **[[3tj6]] - hm-VCL head domain + protein Ps 120 peptide<br /> | ||
| - | **[[4ehp]] - hm-VCL head domain + catenin α-1 residues 277-382<br /> | ||
| - | **[[3vf0]] – hm-VCL residues 856-1134 + ribonucleoprotein PTB-binding<br /> | ||
| - | **[[2gdc]] – cm-VCL Vd1+SfInvasin C-terminal <br /> | ||
| - | **[[1xwj]] - cm-VCL Vd1+cTalin VBS3<br /> | ||
| - | **[[1zvz]], [[1zw2]], [[1zw3]], [[1u6h]] - cm-VCL Vd1+cTalin rod<br /> | ||
| - | **[[1st6]] – cm-VCL<br /> | ||
| - | **[[4e17]], [[4e18]] - cm-VCL Vd1 + catenin α-1 VCL-binding domain | ||
| - | }} | ||
==References== | ==References== | ||
<references /> | <references /> | ||
| + | </StructureSection> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
*Created with the participation of [[User:Susan Craig|Susan Craig]]. | *Created with the participation of [[User:Susan Craig|Susan Craig]]. | ||
Current revision
| |||||||||||
- Created with the participation of Susan Craig.