This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Protein phosphatase
From Proteopedia
(Difference between revisions)
| (28 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
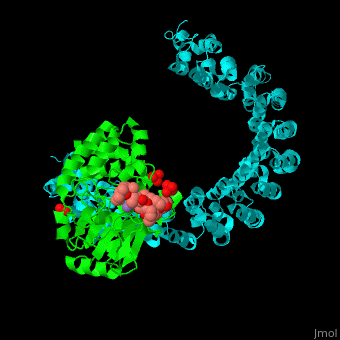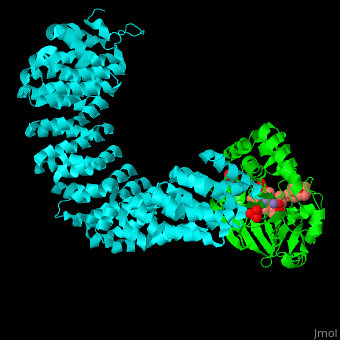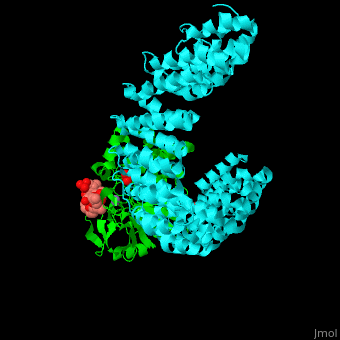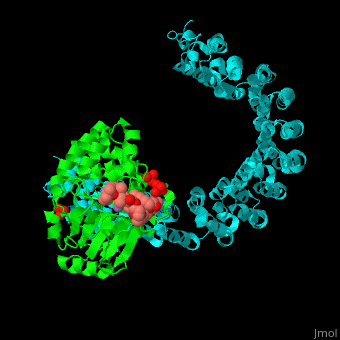
| - | + | <StructureSection load='' size='350' side='right' scene='54/540142/Cv/1' caption='Human PP2A catalytic (green) and regulatory (cyan) subunits complex with tumor-inducing toxin and sulfate [[3k7v]]'> | |
| - | + | __TOC__ | |
| - | + | == Function == | |
| + | '''Protein phosphatases''' (PP) or '''serine/threonine protein phosphatase''' regulate protein phosphorylation and thus are key in intracellular signal transduction processes.<br /> | ||
| + | * '''PP1''' is a serine/threonine phosphatase and is a key component of the insulin signaling pathway<ref>PMID:9609113</ref>.<br /> | ||
| + | * '''PP2A''' targets proteins in the oncogenic signaling pathways<ref>PMID:11812651</ref>. For PP2A see also [[HEAT Repeat]].<br /> | ||
| + | * '''PP2C''' are Mg/Mn-dependent and are essential for the regulation of cell cycle and stress signaling pathways. For details see<br /> | ||
| + | * <scene name='54/540142/Protein_pp2cm_with_mgii/4'>PP2Cm</scene> | ||
| + | *[[Protein Phosphatase 2C]]<br /> | ||
| + | *[[ABA-regulated Protein Phosphatase 2C]]<br /> | ||
| + | *[[ABA Signaling Pathway]].<br /> | ||
| + | * '''PP4''' regulates a variety of cellular functions<ref>PMID:25562660</ref>.<br /> | ||
| + | * '''PP5''' is activated by lipids and is involved in signal transduction<ref>PMID:11137038</ref>. | ||
| + | == Disease == | ||
| + | Mutations in PP2A are found in many solid cancers and leukemias. PP2A-activating drugs are possible candidates for cancer therapeutics protocols<ref>PMID:23639323</ref>. Development of Alzheimer disease drugs could be based on restoration of PP2A activity<ref>PMID:21605044</ref>. | ||
| - | + | == Structural highlights == | |
| - | + | <scene name='54/540142/Cv/5'>Human PP2A catalytic (green) and regulatory (cyan) subunits complex with tumor-inducing toxin</scene>. Algal toxin binds at the surface pocket of the PP2A catalytic subunit which contains the Mn+2 ion cofactors<ref>PMID:19916524</ref>. Water molecule are shown as red sphere. | |
| - | + | *<scene name='54/540142/Cv/6'>Mn+2 ion cofactors coordination site</scene>. | |
| - | + | *<scene name='54/540142/Cv/7'>Whole binding site</scene>. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
== 3D Structures of protein phosphatase== | == 3D Structures of protein phosphatase== | ||
| - | + | [[Protein phosphatase 3D structures]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | [[ | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | </StructureSection> | |
| - | + | == References == | |
| - | + | <references/> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
References
- ↑ Ragolia L, Begum N. Protein phosphatase-1 and insulin action. Mol Cell Biochem. 1998 May;182(1-2):49-58. PMID:9609113
- ↑ Resjo S, Goransson O, Harndahl L, Zolnierowicz S, Manganiello V, Degerman E. Protein phosphatase 2A is the main phosphatase involved in the regulation of protein kinase B in rat adipocytes. Cell Signal. 2002 Mar;14(3):231-8. PMID:11812651
- ↑ Lipinszki Z, Lefevre S, Savoian MS, Singleton MR, Glover DM, Przewloka MR. Centromeric binding and activity of Protein Phosphatase 4. Nat Commun. 2015 Jan 6;6:5894. doi: 10.1038/ncomms6894. PMID:25562660 doi:http://dx.doi.org/10.1038/ncomms6894
- ↑ Chinkers M. Protein phosphatase 5 in signal transduction. Trends Endocrinol Metab. 2001 Jan-Feb;12(1):28-32. PMID:11137038
- ↑ Perrotti D, Neviani P. Protein phosphatase 2A: a target for anticancer therapy. Lancet Oncol. 2013 May;14(6):e229-38. doi: 10.1016/S1470-2045(12)70558-2. PMID:23639323 doi:http://dx.doi.org/10.1016/S1470-2045(12)70558-2
- ↑ Rudrabhatla P, Pant HC. Role of protein phosphatase 2A in Alzheimer's disease. Curr Alzheimer Res. 2011 Sep;8(6):623-32. PMID:21605044
- ↑ Huhn J, Jeffrey PD, Larsen K, Rundberget T, Rise F, Cox NR, Arcus V, Shi Y, Miles CO. A structural basis for the reduced toxicity of dinophysistoxin-2. Chem Res Toxicol. 2009 Nov;22(11):1782-6. PMID:19916524 doi:10.1021/tx9001622
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Alexsandra Tifane Santos do Nascimento, Joel L. Sussman