This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Elongation factor
From Proteopedia
(Difference between revisions)
| (16 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
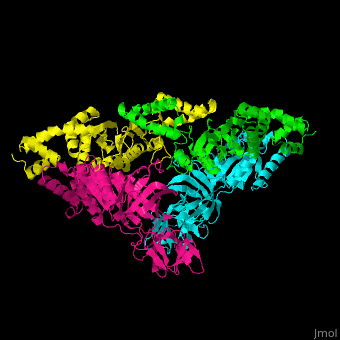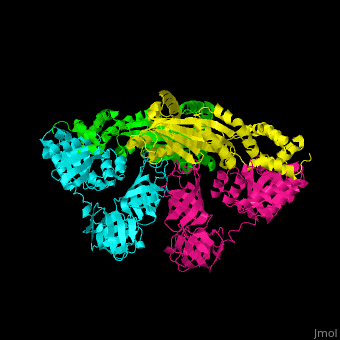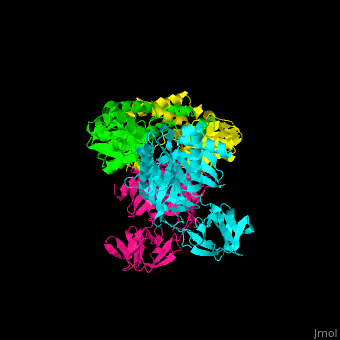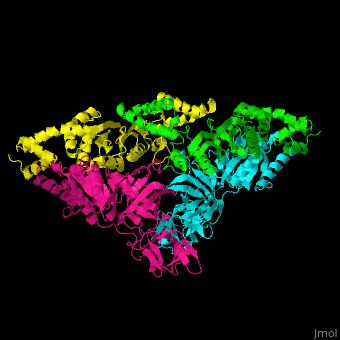
| - | <StructureSection load=' | + | <StructureSection load='' size='350' side='right' caption='Structure of EF-Tu (cyan and magenta) with EF-Ts (green and yellow) (PDB entry [[1efu]])' scene='51/517376/Cv/1'> |
| + | __FORCETOC__ | ||
| + | |||
| + | == Function == | ||
'''Elongation factors''' (EF) facilitate translational elongation during the formation of peptide bonds in the ribosome.<br /> | '''Elongation factors''' (EF) facilitate translational elongation during the formation of peptide bonds in the ribosome.<br /> | ||
| - | * '''EF-selB''' is selenocysteine-specific EF.<br /> | + | * '''EF-selB''' is selenocysteine-specific EF. See [[SelB]]<br /> |
* '''EF-Tu or EF 1-α''' (elongation factor thermo unstable) is a prokaryotic EF. EF-Tu contributes to translational accuracy. It catalyzes the addition of aminoacyl tRNA<ref>PMID:20798060</ref>. <br /> | * '''EF-Tu or EF 1-α''' (elongation factor thermo unstable) is a prokaryotic EF. EF-Tu contributes to translational accuracy. It catalyzes the addition of aminoacyl tRNA<ref>PMID:20798060</ref>. <br /> | ||
*'''EF-Ts or EF 1-β''' (elongation factor thermo stable) catalyzes the release of GDP from EF-Tu.<br /> | *'''EF-Ts or EF 1-β''' (elongation factor thermo stable) catalyzes the release of GDP from EF-Tu.<br /> | ||
| Line 13: | Line 16: | ||
* '''EF-1 γ''' acts during the delivery of aminoacyl tRNA to the ribosome.<br /> | * '''EF-1 γ''' acts during the delivery of aminoacyl tRNA to the ribosome.<br /> | ||
* '''EF-2''' promotes the translocation of the nascent protein chain from the A site to the P site on the ribosome<ref>PMID:16246167</ref>.<br /> | * '''EF-2''' promotes the translocation of the nascent protein chain from the A site to the P site on the ribosome<ref>PMID:16246167</ref>.<br /> | ||
| - | * '''EF-3''' is a unique EF in fungi hence it provides an anti-fungal drug target.<br /> | + | * '''EF-3''' is a unique EF in fungi hence it provides an anti-fungal drug target. See [[HEAT Repeat]]<br /> |
* '''EF Spt4, Spt5, Spt6''' are conserved among eukaryotes. They modulate the chromatin structure.<br /> | * '''EF Spt4, Spt5, Spt6''' are conserved among eukaryotes. They modulate the chromatin structure.<br /> | ||
* '''EF-CA150''' is believed to play a role in coupling transcription and splicing.<br /> | * '''EF-CA150''' is believed to play a role in coupling transcription and splicing.<br /> | ||
| - | * '''Elongin B and C | + | * '''Elongin complex''' or '''SIII''' activates elongation by RNA polymerase II by suppressing transient pausing of the enzyme<ref>PMID:7660129</ref>. The complex is composed of elongin A, B and C. '''Elongin A''' (EloA) is the active component of the complex. '''Elongin B and C''' (EloBC) are the regulatory subunits of it. '''Von Hippel-Landau tumor suppressor protein''' (VHL) binds to EloBC and inhibits transcriptional elongation.<br /> |
| - | + | * '''Negative EF''' (NELF) are involved in regulating the pausing of RNA Pol II polymerase transcripton<ref>PMID:38401543</ref>.<br /> | |
| - | + | ||
| - | </ | + | <scene name='51/517376/Cv/4'>Complex EF-Tu with EF-Ts is heterotetramer</scene>, or, more exactly <scene name='51/517376/Cv/5'>heterodimer of homodimers</scene> (PDB entry [[1efu]]).<ref>PMID:8596629</ref> |
==3D structures of elongation factor== | ==3D structures of elongation factor== | ||
| + | [[Elongation factor 3D structures]] | ||
| - | + | </StructureSection> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | **[[1lqb]], [[1lm8]] – hEloBC + von-Hippel Lindau disease tumor suppressor + hypoxia inducible factor 1 α<br /> | ||
| - | **[[3zrc]], [[3ztc]], [[3ztd]], [[3zun]] - hEloBC + von-Hippel Lindau disease tumor suppressor + inhibitor<br /> | ||
| - | **[[3zrf]] - hEloBC + von-Hippel Lindau disease tumor suppressor<br /> | ||
| - | **[[2c9w]], [[2izv]], [[2jz3]] – hEloBC + suppressor of cytokine signaling<br /> | ||
| - | **[[3dcg]] – hEloBC + virion infectivity factor<br /> | ||
| - | **[[2xai]] - hEloBC + ankyrin rep<br /> | ||
| - | **[[1vcb]] – hEloBC + VHL<br /> | ||
| - | **[[2fnj]] - mEloBC + GUSTAVUS | ||
| - | }} | ||
== References == | == References == | ||
<references/> | <references/> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
References
- ↑ Takeshita D, Tomita K. Assembly of Q{beta} viral RNA polymerase with host translational elongation factors EF-Tu and -Ts. Proc Natl Acad Sci U S A. 2010 Sep 7;107(36):15733-8. Epub 2010 Aug 23. PMID:20798060 doi:http://dx.doi.org/10.1073/pnas.1006559107
- ↑ Jorgensen R, Merrill AR, Andersen GR. The life and death of translation elongation factor 2. Biochem Soc Trans. 2006 Feb;34(Pt 1):1-6. PMID:16246167 doi:http://dx.doi.org/10.1042/BST20060001
- ↑ Aso T, Lane WS, Conaway JW, Conaway RC. Elongin (SIII): a multisubunit regulator of elongation by RNA polymerase II. Science. 1995 Sep 8;269(5229):1439-43. PMID:7660129
- ↑ Su BG, Vos SM. Distinct negative elongation factor conformations regulate RNA polymerase II promoter-proximal pausing. Mol Cell. 2024 Apr 4;84(7):1243-1256.e5. PMID:38401543 doi:10.1016/j.molcel.2024.01.023
- ↑ Kawashima T, Berthet-Colominas C, Wulff M, Cusack S, Leberman R. The structure of the Escherichia coli EF-Tu.EF-Ts complex at 2.5 A resolution. Nature. 1996 Feb 8;379(6565):511-8. PMID:8596629 doi:http://dx.doi.org/10.1038/379511a0