Nitrogenase
From Proteopedia
(Difference between revisions)
| (24 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
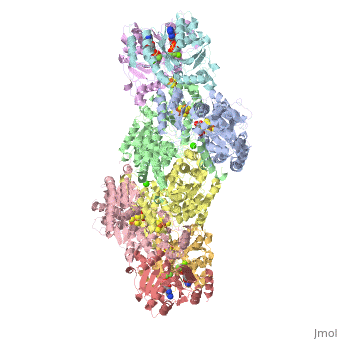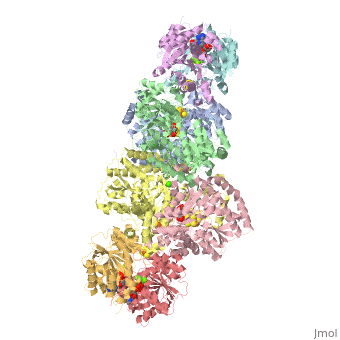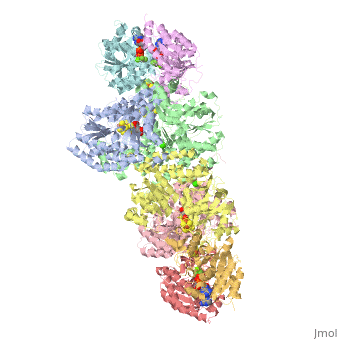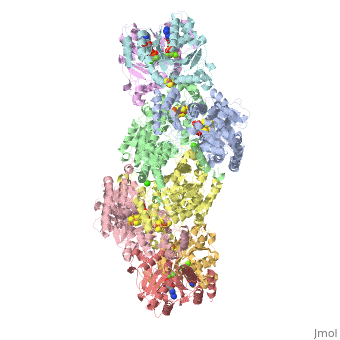
| - | + | <StructureSection load="1N2C" size="350" color="white" caption="Nitrogenase complex: α (grey and green) and β (pink and yellow) chains, nitrogenase iron protein ( purple,cyan, red and gold), showing Fe-Mo-S cluster complex with ADP, adipic acid, AlF4, Ca+2 and Mg+2 ions [[1n2c]]"> | |
| + | __TOC__ | ||
| + | ==Function== | ||
| - | + | '''Nitrogenase''' (Nase) is an enzyme that fixes atmospheric nitrogen (N<sub>2</sub>) into ammonia. Though abundantly present in the atmosphere, most organisms cannot utilize N<sub>2</sub> directly, and must instead take it in through other forms, like ammonia or nitrate. The triple bond in N<sub>2</sub> is highly resistant to changes in oxidation state, and nitrogenases, found only in nitrogen-fixing bacteria, are the only proteins capable of reducing N<sub>2</sub> to ammonia. | |
| - | + | ||
| - | + | ||
| - | '''Nitrogenase''' is an enzyme that fixes atmospheric nitrogen (N<sub>2</sub>) into ammonia. Though abundantly present in the atmosphere, most organisms cannot utilize N<sub>2</sub> directly, and must instead take it in through other forms, like ammonia or nitrate. The triple bond in N<sub>2</sub> is highly resistant to changes in oxidation state, and nitrogenases, found only in nitrogen-fixing bacteria, are the only proteins capable of reducing N<sub>2</sub> to ammonia. | + | |
| Line 20: | Line 19: | ||
[[Image:clip_image002.jpg]] | [[Image:clip_image002.jpg]] | ||
| - | |||
| - | [[Image:nitrogenase]] | ||
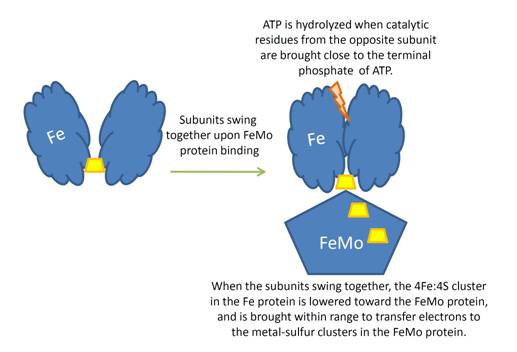
Before this crystal structure showing the complete nitrogenase complex was solved, crystal structures of the Fe and FeMo proteins had been solved, but the mechanisms of ATP hydrolysis and electron transfer were still unknown. In this structure, as indicated in the figure above, the two subunits of the Fe protein were observed to have swung closer together. This movement results from Fe protein binding to FeMo protein. Focusing in on the <scene name='Sandbox_10/1n2c_atp/3'>ATP binding pocket</scene> discussed above, especially on the <scene name='Sandbox_10/1n2c_atp_lys_10/1'>AlF4-</scene> that is an analog for the negatively charged planar transition state reveals that there are several positive charges in this vicinity that stabilize the transition state. Importantly, Lys 10 from the opposite subunit is an important source of stabilizing positive charge. Only in this structure where the two subunits have swung closer together is this residue in position to help catalyze the hydrolysis of the terminal phosphate of ATP. | Before this crystal structure showing the complete nitrogenase complex was solved, crystal structures of the Fe and FeMo proteins had been solved, but the mechanisms of ATP hydrolysis and electron transfer were still unknown. In this structure, as indicated in the figure above, the two subunits of the Fe protein were observed to have swung closer together. This movement results from Fe protein binding to FeMo protein. Focusing in on the <scene name='Sandbox_10/1n2c_atp/3'>ATP binding pocket</scene> discussed above, especially on the <scene name='Sandbox_10/1n2c_atp_lys_10/1'>AlF4-</scene> that is an analog for the negatively charged planar transition state reveals that there are several positive charges in this vicinity that stabilize the transition state. Importantly, Lys 10 from the opposite subunit is an important source of stabilizing positive charge. Only in this structure where the two subunits have swung closer together is this residue in position to help catalyze the hydrolysis of the terminal phosphate of ATP. | ||
| Line 28: | Line 25: | ||
For these reasons, binding of Fe protein to FeMo protein results in hydrolysis of ATP. Additionally, the 4Fe:4S cluster is lowered close enough to the metal-sulfur clusters of the FeMo protein that electron transfer can occur. All three clusters found in the Fe protein-FeMo protein complex can be seen <scene name='Sandbox_10/1n2c_clusters/2'>here</scene>. Once the electrons have passed from the 4Fe:4S cluster of the Fe protein to the 8Fe:7S cluster of the FeMo protein, they then transfer to the 7Fe:Mo:9S:homocitrate:X cluster where X is an unidentified light atom. It is at this cluster where reduction of N<sub>2</sub> and H<sup>+</sup> occur. The exact mechanism of reduction, however, is still unknown. | For these reasons, binding of Fe protein to FeMo protein results in hydrolysis of ATP. Additionally, the 4Fe:4S cluster is lowered close enough to the metal-sulfur clusters of the FeMo protein that electron transfer can occur. All three clusters found in the Fe protein-FeMo protein complex can be seen <scene name='Sandbox_10/1n2c_clusters/2'>here</scene>. Once the electrons have passed from the 4Fe:4S cluster of the Fe protein to the 8Fe:7S cluster of the FeMo protein, they then transfer to the 7Fe:Mo:9S:homocitrate:X cluster where X is an unidentified light atom. It is at this cluster where reduction of N<sub>2</sub> and H<sup>+</sup> occur. The exact mechanism of reduction, however, is still unknown. | ||
| + | ==3D structure of Nitrogenase== | ||
| + | [[Nitrogenase 3D structures]] | ||
| + | </StructureSection> | ||
== References == | == References == | ||
<references/> | <references/> | ||
| - | + | [[Category:Topic Page]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
Current revision
| |||||||||||
References
- ↑ Schindelin H, Kisker C, Schlessman JL, Howard JB, Rees DC. Structure of ADP x AIF4(-)-stabilized nitrogenase complex and its implications for signal transduction. Nature. 1997 May 22;387(6631):370-6. PMID:9163420 doi:10.1038/387370a0
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Alexander Berchansky, Eran Hodis, David Canner, Joel L. Sussman