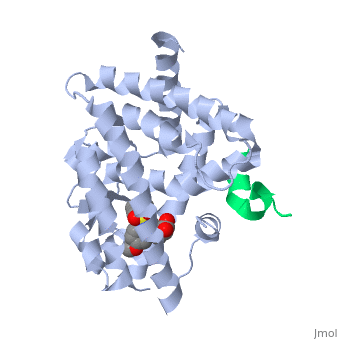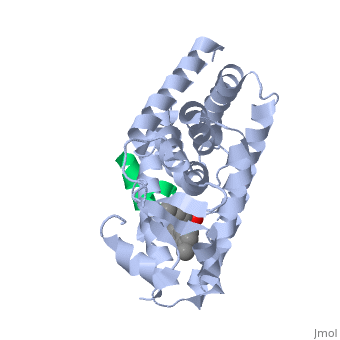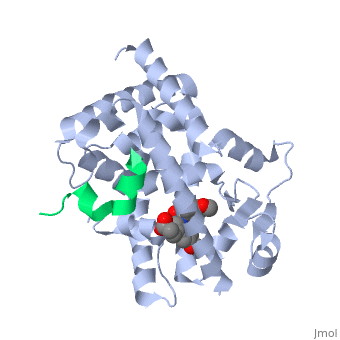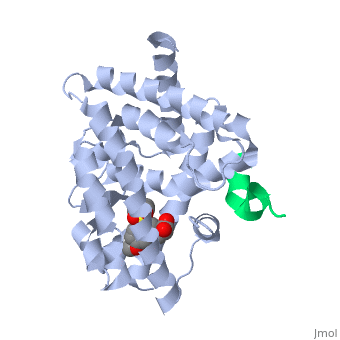
This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
PPAR-gamma
From Proteopedia
| (6 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | < | + | <StructureSection load='3et3' size='450' side='right' scene='' caption='Human PPAR-gamma (grey) complex with NCOA-1 peptide (green) and anti-diabetic agent (PDB code [[3et3]]) '> |
== Introduction == | == Introduction == | ||
Peroxisome proliferator-activated receptor gamma (<scene name='PPAR-gamma/Ppar_gamma/3'>PPAR</scene>γ) is a protein in the nuclear receptors subfamily. It is one of three isotypes (-α, -β/ δ, and -γ) [1] of [[PPAR]] receptors and has two protein isoforms governed by splice variations, which result in differences in the length of the amino (N)-terminal region (PPARγ1 and PPARγ2) [2]. PPARγ is involved in transcriptional regulation of glucose and lipid homeostasis [1], and helps regulate adipocyte differentiation [3]. It has a <scene name='PPAR-gamma/Binding_pocket/1' target='1'>large binding pocket</scene>, which allows it to interact with a wide array of ligands. <scene name='PPAR-gamma/Interacting_residues/3'>Ligand binding</scene> typically triggers a conformational change of PPARγ, notably in the activation function-2 <scene name='PPAR-gamma/Af-2_domain/2'>(AF-2) domain</scene>, which aids in the recruitment of co-regulatory factors to regulate gene transcription. PPARγ can form a <scene name='PPAR-gamma/Ppar_rxr/3'>heterodimer</scene> with retinoic X receptor alpha (RXRα), a process necessary for most PPARγ-DNA interactions [4]. PPARγ is a molecular target for antidiabetic drugs such as thiazolidinediones (TZDs), which makes the protein a target for Type II Diabetes (T2D) drug research. Due to its involvement in metabolic and inflammatory processes, PPARγ also holds potential for treatments of many metabolic and chronic-inflammatory diseases, such as metabolic syndrome and inflammatory bowel disease, respectively. Errors in PPARγ-related regulation have also been implicated in atherosclerosis and various cancers, like colorectal, breast, and prostate cancers. | Peroxisome proliferator-activated receptor gamma (<scene name='PPAR-gamma/Ppar_gamma/3'>PPAR</scene>γ) is a protein in the nuclear receptors subfamily. It is one of three isotypes (-α, -β/ δ, and -γ) [1] of [[PPAR]] receptors and has two protein isoforms governed by splice variations, which result in differences in the length of the amino (N)-terminal region (PPARγ1 and PPARγ2) [2]. PPARγ is involved in transcriptional regulation of glucose and lipid homeostasis [1], and helps regulate adipocyte differentiation [3]. It has a <scene name='PPAR-gamma/Binding_pocket/1' target='1'>large binding pocket</scene>, which allows it to interact with a wide array of ligands. <scene name='PPAR-gamma/Interacting_residues/3'>Ligand binding</scene> typically triggers a conformational change of PPARγ, notably in the activation function-2 <scene name='PPAR-gamma/Af-2_domain/2'>(AF-2) domain</scene>, which aids in the recruitment of co-regulatory factors to regulate gene transcription. PPARγ can form a <scene name='PPAR-gamma/Ppar_rxr/3'>heterodimer</scene> with retinoic X receptor alpha (RXRα), a process necessary for most PPARγ-DNA interactions [4]. PPARγ is a molecular target for antidiabetic drugs such as thiazolidinediones (TZDs), which makes the protein a target for Type II Diabetes (T2D) drug research. Due to its involvement in metabolic and inflammatory processes, PPARγ also holds potential for treatments of many metabolic and chronic-inflammatory diseases, such as metabolic syndrome and inflammatory bowel disease, respectively. Errors in PPARγ-related regulation have also been implicated in atherosclerosis and various cancers, like colorectal, breast, and prostate cancers. | ||
| + | |||
| + | See also [[Intracellular receptors]] | ||
== Overall Structure and Ligand Binding == | == Overall Structure and Ligand Binding == | ||
| Line 9: | Line 11: | ||
PPARγ is composed of the ligand-independent activation domain (AF-1 region and A/B-domain), a DNA-binding domain (DBD) (C-domain), a hinge region (D-domain), and a ligand-dependent ligand-binding domain (LBD) (E/F-domain and AF-2 region) [5]. The two PPARγ isoforms, PPARγ1 and PPARγ2, differ by only 30 amino acids at the N-terminal end. These added amino acids on PPARγ2 result in increased potency and adipose-selectivity, which makes this protein a key player of adipocyte differentiation [3]. | PPARγ is composed of the ligand-independent activation domain (AF-1 region and A/B-domain), a DNA-binding domain (DBD) (C-domain), a hinge region (D-domain), and a ligand-dependent ligand-binding domain (LBD) (E/F-domain and AF-2 region) [5]. The two PPARγ isoforms, PPARγ1 and PPARγ2, differ by only 30 amino acids at the N-terminal end. These added amino acids on PPARγ2 result in increased potency and adipose-selectivity, which makes this protein a key player of adipocyte differentiation [3]. | ||
The <scene name='PPAR-gamma/Lbd/2' target='2'>ligand binding domain</scene> is composed of 13 α helices and 4 short β strands [1]. It has a T-shaped binding pocket with a volume of ~1440 Å3 [1, 6], which is larger than that of most nuclear receptors [7], allowing for interactions with a variety of ligands [8]. The PPARγ LBD is folded into a helical sandwich to provide a binding site for ligands. It is located at the C-terminal end of PPARγ and is composed of about 250 amino acids [5]. Activation by full agonists occurs through hydrogen bond interactions between the S289, H323, Y473, and H449 residues of the PPARγ-LBD [7] and polar functional groups on the ligand which are typically carbonyl or carboxyl oxygen atoms. Agonist binding results in a conformational change of the LBD AF-2 region, which is necessary for coactivator recruitment. This change can either be dramatic or subtle [1], which leads to stabilization of a charge clamp between helices H3 and H12 [9] to aid in associations with the LXXLL (L, leucine; X, any amino acid) motif of the coactivator [1, 10]. Ligand binding of PPARγ is regulated by communication between the N-terminal A/B domain, which is adjacent to the DBD, and the carboxyl-terminal LBD [11]. | The <scene name='PPAR-gamma/Lbd/2' target='2'>ligand binding domain</scene> is composed of 13 α helices and 4 short β strands [1]. It has a T-shaped binding pocket with a volume of ~1440 Å3 [1, 6], which is larger than that of most nuclear receptors [7], allowing for interactions with a variety of ligands [8]. The PPARγ LBD is folded into a helical sandwich to provide a binding site for ligands. It is located at the C-terminal end of PPARγ and is composed of about 250 amino acids [5]. Activation by full agonists occurs through hydrogen bond interactions between the S289, H323, Y473, and H449 residues of the PPARγ-LBD [7] and polar functional groups on the ligand which are typically carbonyl or carboxyl oxygen atoms. Agonist binding results in a conformational change of the LBD AF-2 region, which is necessary for coactivator recruitment. This change can either be dramatic or subtle [1], which leads to stabilization of a charge clamp between helices H3 and H12 [9] to aid in associations with the LXXLL (L, leucine; X, any amino acid) motif of the coactivator [1, 10]. Ligand binding of PPARγ is regulated by communication between the N-terminal A/B domain, which is adjacent to the DBD, and the carboxyl-terminal LBD [11]. | ||
| - | |||
| - | <applet load='2f4b' size='300' frame='true' align='left' caption='PPARγ Ligand Binding Domain' name='2' /> | ||
== Ligand Activity == | == Ligand Activity == | ||
| Line 33: | Line 33: | ||
PPARγ is found in high levels in colonic epithelial cells. The role of PPARγ in these cells may be related to regulation of immune response and colon inflammation [12]. The onset of Inflammatory Bowel Disease is thought to be caused by inflammatory cytokines present in the colon [12]. In patients with ulcerative colitis, colonic epithelial cells displayed impaired expression of PPARγ, an important mediator of aminosalicylate activities in Inflammatory Bowel Diseases [13]. TZD ligands could be implemented to reduce colonic inflammation [12]. Agonists have also been used in the treatment of colitis and psoriasis by inhibiting the inflammatory response of the epithelium and reducing cytokine production [8]. PPARγ inhibits activity of nuclear factor NFκB, which is higher in active ulcerative colitis patients [15]. | PPARγ is found in high levels in colonic epithelial cells. The role of PPARγ in these cells may be related to regulation of immune response and colon inflammation [12]. The onset of Inflammatory Bowel Disease is thought to be caused by inflammatory cytokines present in the colon [12]. In patients with ulcerative colitis, colonic epithelial cells displayed impaired expression of PPARγ, an important mediator of aminosalicylate activities in Inflammatory Bowel Diseases [13]. TZD ligands could be implemented to reduce colonic inflammation [12]. Agonists have also been used in the treatment of colitis and psoriasis by inhibiting the inflammatory response of the epithelium and reducing cytokine production [8]. PPARγ inhibits activity of nuclear factor NFκB, which is higher in active ulcerative colitis patients [15]. | ||
PPARγ could also be implemented in the treatment of other chronic inflammation-related diseases. Immunomodulatory effects have been found with PPARγ agonists [16]. Rosiglitazone alongside adiponectin reduces renal disease, atherosclerosis, and production of autoantibodies, all of which are characteristic of the inflammatory autoimmune disease Systemic Lupus Erythematosus (SLE) [16]. PPARγ ligands hold potential as cancer treatments [11] due to their ability to inhibit angiogenesis, the process required for the growth and metastasis of solid tumors [8]. PPARγ activators have pro-differentiation and anti-proliferation effects [3]. TZDs have also been shown to inhibit proliferation of human breast, prostate, and colon cancer cells [8]. | PPARγ could also be implemented in the treatment of other chronic inflammation-related diseases. Immunomodulatory effects have been found with PPARγ agonists [16]. Rosiglitazone alongside adiponectin reduces renal disease, atherosclerosis, and production of autoantibodies, all of which are characteristic of the inflammatory autoimmune disease Systemic Lupus Erythematosus (SLE) [16]. PPARγ ligands hold potential as cancer treatments [11] due to their ability to inhibit angiogenesis, the process required for the growth and metastasis of solid tumors [8]. PPARγ activators have pro-differentiation and anti-proliferation effects [3]. TZDs have also been shown to inhibit proliferation of human breast, prostate, and colon cancer cells [8]. | ||
| + | </StructureSection> | ||
| + | __NOTOC__ | ||
| + | ==3D structures of PPAR== | ||
| + | |||
| + | [[Peroxisome Proliferator-Activated Receptors]] | ||
==Additional Resources== | ==Additional Resources== | ||
Current revision
| |||||||||||
3D structures of PPAR
Peroxisome Proliferator-Activated Receptors
Additional Resources
For additional information See: Diabetes
For additional information See: Regulation of Gene Expression
For additional information See: Peroxisome Proliferator-Activated Receptors
References
[1] Gampe Jr RT, Montana VG, Lambert MH, et al. Asymmetry in the PPARγ/RXRα crystal structure reveals the molecular basis of heterodimerization among nuclear receptors (2000) Molecular Cell, 15(9), pp.545-555.
[2] Zieleniak A, Wójcik M, Woźniak LA. Structure and physiology functions of the human peroxisome proliferator-activated receptor γ (2008) Arch. Immunol. Ther. Exp., 56 (5), pp. 331-345.
[3] Tontonoz P, Spiegelman BM. Fat and Beyond: The Diverse Biology of PPARγ (2008) Annu. Rev. Biochem., 77, pp. 289-312.
[4] Desvergne B, Wahli W. Peroxisome proliferator-activated receptors: Nuclear control of metabolism (1999) Endocrine Reviews, 20 (5), pp. 649-688.
[5] Lewis SN, Bassaganya-Riera J, Bevan DR. Virtual Screening as a Technique for PPAR Modulatory Discovery (2010) PPAR Research, 2010, pp. 861238.
[6] Itoh T, Fairall L, Amin K, et al. Structural basis for the activation of PPARγ by oxidized fatty acids (2008) Nature Structural and Molecular Biology, 15 (9), pp.924-931.
[7] Pochetti G, Godio C, Mitro N, et al. Insights into the mechanism of partial agonism: crystal structures of the peroxisome proliferator-activated receptor γ ligand-binding domain in the complex with two enantiomeric ligands (2007) Journal of Biological Chemistry, 282 (23), pp.17314-17324.
[8] Murphy GJ, Holder JC. PPAR-γ agonists: Therapeutic role in diabetes, inflammation and cancer (2000) Trends in Pharmacological Sciences, 21 (12), pp. 469-474.
[9] Xu HE, Stanley TB, Montana VG, et al. Structural basis for antagonist-mediated recruitment of nuclear co-repressors by PPARα (2002) Nature, 415 (6873), pp.813-817.
[10] Kallenberger BC, Love, JD, Chatterjee VKK, Schwabe JWR. A dynamic mechanism of nuclear receptor activation and its perturbation in a human disease (2003) Nature, 10 (2), pp.136-140.
[11] Shao D, Rangwala SM, Bailey ST, Krakow SA, Reginato MJ, Lazar MA. Interdomain communication regulating ligand binding by PPARγ (1998) Nature, 396, pp. 377-380.
[12] Su CG, Wen X, Bailey ST, Jiang W, Rangwala SM, Keilbaugh SA, Flanigan A, Murthy S, Lazar MA, Wu GD. A Novel therapy for colitis utilizing PPAR-γ ligands to inhibit the epithelial inflammatory response (1999) J Clin Invest., 104(4), pp. 383-389.
[13] Dubuquoy L, Rousseaux C, Thuru X, Peyrin-Biroulet L, Romano O, Chavatte P, Chamaillard M, Desreumaux P. PPARγ as a new therapeutic target in inflammatory bowel disease (2006) International Journal of Gastroenterology and Hepatology, 55 (9), pp.1341-1349.
[14] McKenna NJ, O'Malley BW. Combinatorial control of gene expression by nuclear receptors and coregulators (2002) Cell, 108 (4), pp. 465-474.
[15] Sartor, RB. Mechanisms of disease: pathogenesis of Crohn’s disease and ulcerative colitis (2006) Nature, 3(7), pp. 390-407.
[16] Aprahamian T, Bonegio RG, Richez C, Yasuda K, Chiang L, Sato K, Walsh K, Rifkin IR. The Peroxisome Proliferator-Activated Receptor γ Agonist Rosiglitazone Ameliorates Murine Lupus by Induction of Adiponectin (2009) the Journal of Immunology, 182, pp. 340 -346.
Proteopedia Page Contributors and Editors (what is this?)
Michal Harel, Lera Brannan, David Canner, Alexander Berchansky