This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Mannosidase
From Proteopedia
(Difference between revisions)
| (22 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
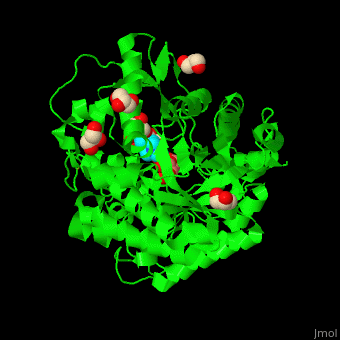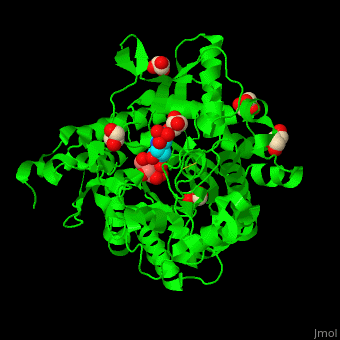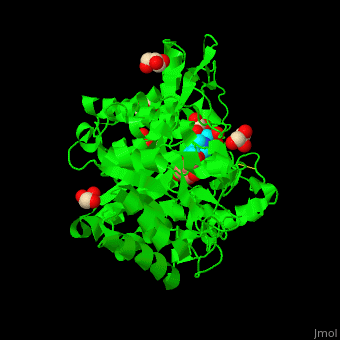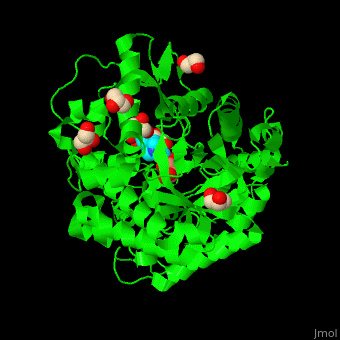
| - | + | <StructureSection load='4jie' size='350' side='right' caption='Rice β-mannosidase complex with β-mannose, glycerol and HEPES (PDB entry [[4jie]])' scene='45/458458/Cv/1'> | |
| + | __TOC__ | ||
| + | == Function == | ||
| - | '''Mannosidase''' (MAN) is an enzyme which hydrolyzes mannose. There are 2 kinds of MAN. | + | '''Mannosidase''' (MAN) is an enzyme which hydrolyzes mannose. There are 2 kinds of MAN. '''α-MAN''' which hydrolyzes α-mannose<ref>PMID:12634058</ref> and '''β-MAN''' which hydrolyzes β-1,4-linked manno-oligosaccharides<ref>PMID:10553664</ref>. α-MAN isozymes are classified as class I and II. '''Mannan endo-1,4-β-MAN''' hydrolyzes 1-4-β-D-mannosidic linkages in mannans, galactomannans and glucomannans. See some details in [[Molecular Playground/ERMan1]]. See also [[Carbohydrate Metabolism]]. |
| - | + | == Disease == | |
| + | α-mannosidosis is manifested by accumulation of mannose-containing oligosaccharides leading to mental retardation, hearing impairment, skeletal changes and immunodeficiency<ref>PMID:9915946</ref>. β-MAN deficiency called β-mannosidosis is a disorder of oligosaccharide metabolism<ref>PMID:12890191</ref>. | ||
| + | |||
| + | == Structural highlights == | ||
| + | <scene name='45/458458/Cv/5'>The active site of β-MAN contains β-mannose</scene><ref>PMID:24100330</ref>. Water molecules shown as red spheres. | ||
==3D structures of mannosidase== | ==3D structures of mannosidase== | ||
| + | [[Mannosidase 3D structures]] | ||
| - | + | </StructureSection> | |
| - | + | == References == | |
| - | + | <references/> | |
| - | ===α-MAN class I=== | ||
| - | |||
| - | [[1fmi]] - hαMAN I - human<br /> | ||
| - | [[1fo2]] - hαMAN I + deoxymannojirimycin<br /> | ||
| - | [[1fo3]] - hαMAN I+ kifunensine<br /> | ||
| - | [[1dl2]] - αMAN I catalytic domain + mannose - yeast<br /> | ||
| - | [[2ri8]] - PcαMAN I + glycerol – ''Penicillium citrinum''<br /> | ||
| - | [[1kre]], [[1kkt]] - PcαMAN I + mannose<br /> | ||
| - | [[1krf]] - PcαMAN I + kifunensine + mannose<br /> | ||
| - | [[1x9d]] - hαMAN I residues 243-699 + polysaccharide analog<br /> | ||
| - | [[1nxc]] - αMAN I + mannose – mouse<br /> | ||
| - | [[1g6i]] - αMAN I + deoxymannojirimycin<br /> | ||
| - | |||
| - | ===α-MAN class II=== | ||
| - | |||
| - | [[2wyh]] - SpαMAN II – ''Streptococcus pyrogenes''<br /> | ||
| - | [[3bub]], [[1hty]] - DmαMAN II catalytic domain – ''Drosophila melanogaster''<br /> | ||
| - | [[3bud]] - DmαMAN II catalytic domain (mutant) <br /> | ||
| - | [[3bui]] - DmαMAN II catalytic domain (mutant) + Tris<br /> | ||
| - | [[3bup]], [[3buq]], [[3czs]] - DmαMAN II catalytic domain (mutant) + mannose<br /> | ||
| - | [[3bvt]], [[3bvu]], [[3bvv]], [[3bvw]], [[3bvx]] - DmαMAN II catalytic domain (mutant) + mannopyranoside<br /> | ||
| - | [[1r33]], [[1r34]] - DmαMAN II catalytic domain + mannopyranoside<br /> | ||
| - | [[3ddf]], [[3ddg]], [[2f18]], [[2f1a]], [[2f1b]] - DmαMAN II catalytic domain + pyrrolidin<br /> | ||
| - | [[2alw]] - DmαMAN II catalytic domain + noeuromycin<br /> | ||
| - | [[3cv5]] - DmαMAN II catalytic domain + polysaccharide<br /> | ||
| - | [[3czn]] - DmαMAN II catalytic domain (mutant) + polysaccharide<br /> | ||
| - | [[2wyi]] - SpαMAN II + swainsonine<br /> | ||
| - | [[3blb]], [[1hww]] - DmαMAN II + swainsonine<br /> | ||
| - | [[3ejp]], [[3ejq]], [[3ejr]], [[3ejs]], [[3ejt]], [[3eju]], [[2ow6]], [[2ow7]] - DmαMAN II catalytic domain + swainsonine analog<br /> | ||
| - | [[3dx0]], [[3dx2]], [[2f7o]] - DmαMAN II catalytic domain + mannostatine<br /> | ||
| - | [[3dx1]], [[3dx3]], [[3dx4]], [[2f7p]] - DmαMAN II catalytic domain + mannostatine analog<br /> | ||
| - | [[3d4y]], [[3d4z]] - DmαMAN II catalytic domain + imidazole derivative<br /> | ||
| - | [[3d50]], [[3d51]], [[3d52]] - DmαMAN II catalytic domain + inhibitor<br /> | ||
| - | [[2fyv]], [[2f7q]], [[2f7r]] - DmαMAN II catalytic domain + amino-salacinol carboxylate analog<br /> | ||
| - | [[1tqv]], [[1tqw]], [[1tqs]], [[1tqt]], [[1tqu]] - DmαMAN II catalytic domain (mutant) + seleno-salacinol<br /> | ||
| - | [[1ps3]] - DmαMAN II catalytic domain + kifunensine<br /> | ||
| - | [[1qwn]] - DmαMAN II catalytic domain + intermediate<br /> | ||
| - | [[1hxk]] - DmαMAN II catalytic domain + deoxymannojirimycin<br /> | ||
| - | [[1qwu]] - DmαMAN II catalytic domain (mutant) + guloside<br /> | ||
| - | [[1qx1]] - DmαMAN II catalytic domain (mutant) + mannosyl | ||
| - | |||
| - | ===β-MAN=== | ||
| - | |||
| - | [[2je8]] - BtβMAN residues 26-864 – ''Bacterioides thetaiotaomicron''<br /> | ||
| - | [[2wbk]] - BtβMAN residues 26-864 + mannopyranoside <br /> | ||
| - | [[2vqt]], [[2vr4]], [[2vjx]], [[2vl4]], [[2vmf]], [[2vo5]], [[2vot]], [[2vqu]] - BtβMAN residues 26-864 + piperidine derivative<br /> | ||
| - | [[1oh4]], [[1of4]] - TmβMAN catalytic domain + mannose – ''Thermotoga maritima''<br /> | ||
| - | [[1of3]] - TmβMAN catalytic domain<br /> | ||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
| - | |||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
References
- ↑ Heikinheimo P, Helland R, Leiros HK, Leiros I, Karlsen S, Evjen G, Ravelli R, Schoehn G, Ruigrok R, Tollersrud OK, McSweeney S, Hough E. The structure of bovine lysosomal alpha-mannosidase suggests a novel mechanism for low-pH activation. J Mol Biol. 2003 Mar 28;327(3):631-44. PMID:12634058
- ↑ Ademark P, Lundqvist J, Hagglund P, Tenkanen M, Torto N, Tjerneld F, Stalbrand H. Hydrolytic properties of a beta-mannosidase purified from Aspergillus niger. J Biotechnol. 1999 Oct 8;75(2-3):281-9. PMID:10553664
- ↑ Berg T, Riise HM, Hansen GM, Malm D, Tranebjaerg L, Tollersrud OK, Nilssen O. Spectrum of mutations in alpha-mannosidosis. Am J Hum Genet. 1999 Jan;64(1):77-88. PMID:9915946 doi:http://dx.doi.org/S0002-9297(07)61660-7
- ↑ Uchino Y, Fukushige T, Yotsumoto S, Hashiguchi T, Taguchi H, Suzuki N, Konohana I, Kanzaki T. Morphological and biochemical studies of human beta-mannosidosis: identification of a novel beta-mannosidase gene mutation. Br J Dermatol. 2003 Jul;149(1):23-9. PMID:12890191
- ↑ Tankrathok A, Iglesias-Fernandez J, Luang S, Robinson RC, Kimura A, Rovira C, Hrmova M, Ketudat Cairns JR. Structural analysis and insights into the glycon specificity of the rice GH1 Os7BGlu26 beta-D-mannosidase. Acta Crystallogr D Biol Crystallogr. 2013 Oct;69(Pt 10):2124-35. doi:, 10.1107/S0907444913020568. Epub 2013 Sep 20. PMID:24100330 doi:http://dx.doi.org/10.1107/S0907444913020568