This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Subtilisin
From Proteopedia
(Difference between revisions)
| (26 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
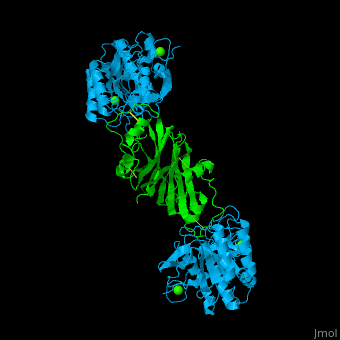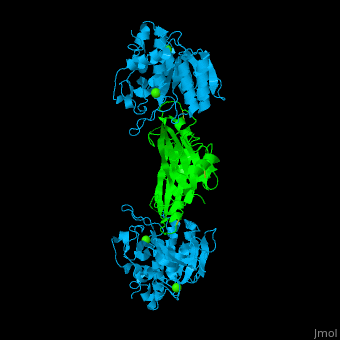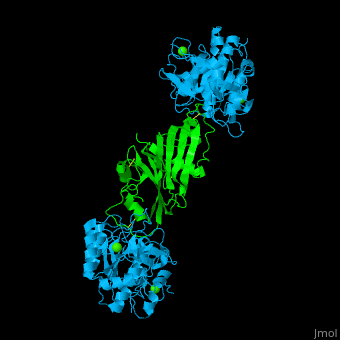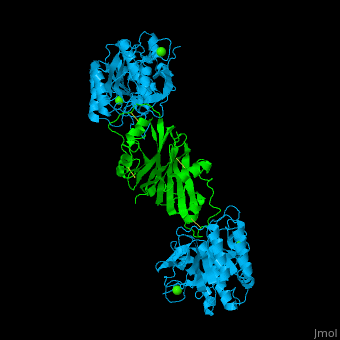
| - | + | <StructureSection load='' size='350' side='right' caption='Subtilisin (deepskyblue) complex with streptomycin inhibitor (green) and Ca+2 ions (green) (PDB entry [[2sic]])' scene='43/430882/Cv/2'> | |
| - | + | == Function == | |
| + | [[Subtilisin]] is a serine protease. A 77 amino acid propeptide is cleaved from the N-terminus of pro-Sub to create the mature active enzym<ref>PMID:4967581</ref>. See detalis in [[User:Tommie Hata/Introduction to Protein Engineering-Subtilisin]]. | ||
| - | + | *'''Selenosubtilisin''' is a semisynthetic selenoenzyme produced by chemical modification of subtilisin<ref>PMID:8385489</ref>.<br /> | |
| - | + | *'''Savinase''' is a subtilisin produced from a lentil<ref>PMID:24738747</ref>.<br /> | |
| - | + | == Relevance == | |
| + | Subtilisin is widely used commercially in detergents. | ||
| + | |||
| + | == Structural highlights == | ||
| + | The active site of Sub contains the <scene name='43/430882/Cv/12'>catalytic triad: Ser-His-Asp</scene>. The <scene name='43/430882/Cv/13'>peptide inhibitor has numerous interactions with Sub</scene> and the <scene name='43/430882/Cv/14'>scissile bond is flanked by Cys-Pro-Met-Val</scene><ref>PMID:1920411</ref>. | ||
== 3D Structures of Subtilisin == | == 3D Structures of Subtilisin == | ||
| + | [[Subtilisin 3D structures]] | ||
| - | + | </StructureSection> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | == References == | ||
| + | <references/> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
References
- ↑ Smith EL, DeLange RJ, Evans WH, Landon M, Markland FS. Subtilisin Carlsberg. V. The complete sequence; comparison with subtilisin BPN'; evolutionary relationships. J Biol Chem. 1968 May 10;243(9):2184-91. PMID:4967581
- ↑ Bell IM, Fisher ML, Wu ZP, Hilvert D. Kinetic studies on the peroxidase activity of selenosubtilisin. Biochemistry. 1993 Apr 13;32(14):3754-62. PMID:8385489
- ↑ Garcia-Mora P, Penas E, Frias J, Martinez-Villaluenga C. Savinase, the most suitable enzyme for releasing peptides from lentil (Lens culinaris var. Castellana) protein concentrates with multifunctional properties. J Agric Food Chem. 2014 May 7;62(18):4166-74. doi: 10.1021/jf500849u. Epub 2014, Apr 28. PMID:24738747 doi:http://dx.doi.org/10.1021/jf500849u
- ↑ Takeuchi Y, Satow Y, Nakamura KT, Mitsui Y. Refined crystal structure of the complex of subtilisin BPN' and Streptomyces subtilisin inhibitor at 1.8 A resolution. J Mol Biol. 1991 Sep 5;221(1):309-25. PMID:1920411