This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Transferrin receptor
From Proteopedia
| (17 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
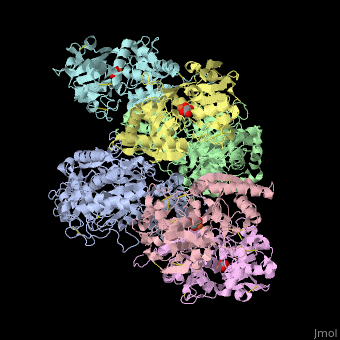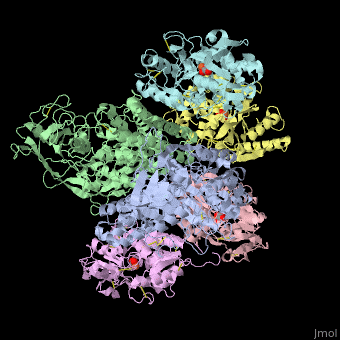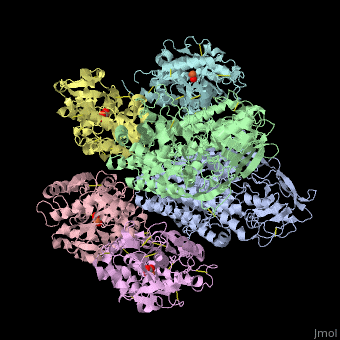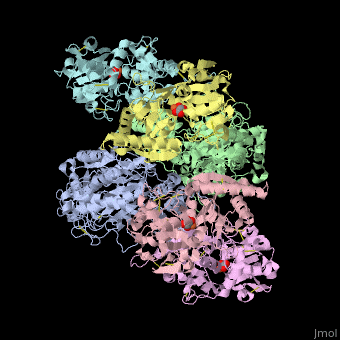
| - | + | <StructureSection load='' size='340' side='right' caption='Human transferrin receptor ectodomain dimer (grey and green) complex with transferrin N-lobe (pink and yellow) and C-lobe (cyan and magenta) and carbonate, [[1suv]]' scene='48/485628/Cv/1' > | |
| - | + | == Function == | |
| - | + | '''Transferrin receptor''' (TfR) imports iron into the cell by receptor-mediated endocytosis of [[Transferrin|transferrin]]-iron complex. TfR is regulated by intracellular iron concentration. The amount of TfR expressed on the cell is proportional to the cell’s need for iron. The TfR has selective affinity for diferric-transferrin. The TfR-transferrin complex is internalized by the cell and iron is released to the cytosol. The TfR-apotransferrin complex returns to the extracellular surface where the apotransferrin dissociates and is replaced by diferric-transferrin<ref>PMID:10192390</ref>. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | '''Transferrin receptor''' (TfR) imports iron into the cell by receptor-mediated endocytosis of transferrin-iron complex. TfR is regulated by intracellular iron concentration. The amount of TfR expressed on the cell is proportional to the cell’s need for iron. The TfR has selective affinity for diferric-transferrin. The TfR-transferrin complex is internalized by the cell and iron is released to the cytosol. The TfR-apotransferrin complex returns to the extracellular surface where the apotransferrin dissociates and is replaced by diferric-transferrin. | + | |
| + | == Relevance == | ||
| + | TfR is a potential determinant of iron loading in congenital anemias<ref>PMID:10468869</ref>. | ||
| + | == Structural highlights == | ||
| + | *<scene name='48/485628/Cv/3'>Human transferrin receptor ectodomain dimer complex with transferrin N-lobe and C-lobe</scene> ([[1suv]]). | ||
| + | </StructureSection> | ||
==3D structures of transferrin receptor== | ==3D structures of transferrin receptor== | ||
| + | Updated on {{REVISIONDAY2}}-{{MONTHNAME|{{REVISIONMONTH}}}}-{{REVISIONYEAR}} | ||
| + | |||
| + | Domains – apical 197-378; ectodomain 121-760 | ||
[[1cx8]] – hTfR ectodomain – human<br /> | [[1cx8]] – hTfR ectodomain – human<br /> | ||
| + | [[6y76]], [[8p0z]] - hTfR apical domain <br /> | ||
[[1de4]] - hTfR ectodomain + hemochromatosis protein + β-2-microglobulin<br /> | [[1de4]] - hTfR ectodomain + hemochromatosis protein + β-2-microglobulin<br /> | ||
| - | [[1suv]] - hTfR ectodomain + transferrin<br /> | + | [[1suv]], [[6soy]], [[6soz]] - hTfR ectodomain + transferrin<br /> |
| - | [[3s9l]], [[3s9m]], [[3s9n]]- hTfR ectodomain + transferrin N-lobe<br /> | + | [[6okd]] - hTfR ectodomain + TfR-binding cysteine-dense peptide<br /> |
| - | [[2nsu]] - hTfR ectodomain in dog retrovirus – EM<br /> | + | [[6d03]], [[6d04]], [[6d05]] - hTfR ectodomain + transferrin – Cryo EM<br /> |
| - | [[3kas]] - hTfR ectodomain + Machupo virus GP1 | + | [[6gsr]], [[6h5i]] - hTfR ectodomain + ferritin heavy chain – Cryo EM<br /> |
| + | [[6s9j]] - hTfR ectodomain + PRE-GP-C<br /> | ||
| + | [[3s9l]], [[3s9m]], [[3s9n]] - hTfR ectodomain + transferrin N-lobe<br /> | ||
| + | [[2nsu]] - hTfR ectodomain in dog retrovirus – Cryo EM<br /> | ||
| + | [[3kas]] - hTfR ectodomain + Machupo virus GP1<br /> | ||
| + | [[6wrv]], [[6wrw]], [[6wrx]] - hTfR ectodomain + synthetic protein<br /> | ||
| + | [[7zqs]] - hTfR + DNA – Cryo EM<br /> | ||
| + | == References == | ||
| + | <references/> | ||
| + | |||
| + | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
3D structures of transferrin receptor
Updated on 22-January-2024
Domains – apical 197-378; ectodomain 121-760
1cx8 – hTfR ectodomain – human
6y76, 8p0z - hTfR apical domain
1de4 - hTfR ectodomain + hemochromatosis protein + β-2-microglobulin
1suv, 6soy, 6soz - hTfR ectodomain + transferrin
6okd - hTfR ectodomain + TfR-binding cysteine-dense peptide
6d03, 6d04, 6d05 - hTfR ectodomain + transferrin – Cryo EM
6gsr, 6h5i - hTfR ectodomain + ferritin heavy chain – Cryo EM
6s9j - hTfR ectodomain + PRE-GP-C
3s9l, 3s9m, 3s9n - hTfR ectodomain + transferrin N-lobe
2nsu - hTfR ectodomain in dog retrovirus – Cryo EM
3kas - hTfR ectodomain + Machupo virus GP1
6wrv, 6wrw, 6wrx - hTfR ectodomain + synthetic protein
7zqs - hTfR + DNA – Cryo EM
References
- ↑ Levy JE, Jin O, Fujiwara Y, Kuo F, Andrews NC. Transferrin receptor is necessary for development of erythrocytes and the nervous system. Nat Genet. 1999 Apr;21(4):396-9. PMID:10192390 doi:http://dx.doi.org/10.1038/7727
- ↑ Cazzola M, Beguin Y, Bergamaschi G, Guarnone R, Cerani P, Barella S, Cao A, Galanello R. Soluble transferrin receptor as a potential determinant of iron loading in congenital anaemias due to ineffective erythropoiesis. Br J Haematol. 1999 Sep;106(3):752-5. PMID:10468869