We apologize for Proteopedia being slow to respond. For the past two years, a new implementation of Proteopedia has been being built. Soon, it will replace this 18-year old system. All existing content will be moved to the new system at a date that will be announced here.
1qki
From Proteopedia
(Difference between revisions)
(New page: 200px <!-- The line below this paragraph, containing "STRUCTURE_1qki", creates the "Structure Box" on the page. You may change the PDB parameter (which sets the PD...) |
|||
| (9 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | [[Image:1qki.png|left|200px]] | ||
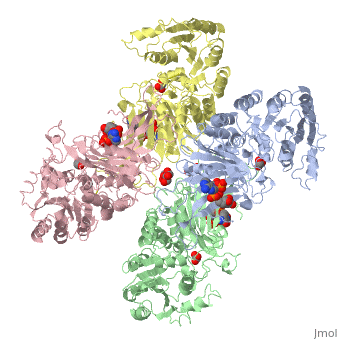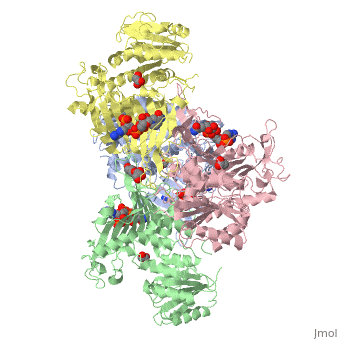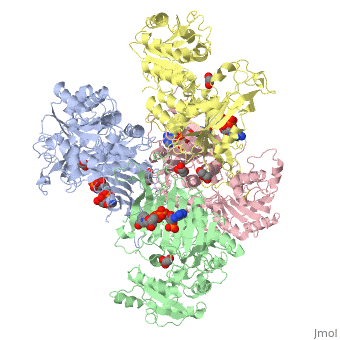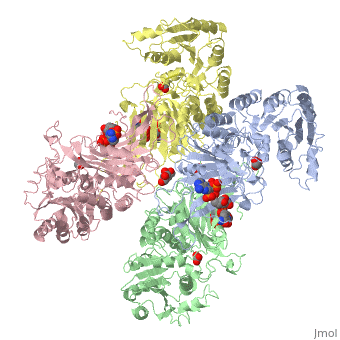
| - | < | + | ==X-RAY STRUCTURE OF HUMAN GLUCOSE 6-PHOSPHATE DEHYDROGENASE (VARIANT CANTON R459L) COMPLEXED WITH STRUCTURAL NADP+== |
| - | + | <StructureSection load='1qki' size='340' side='right'caption='[[1qki]], [[Resolution|resolution]] 3.00Å' scene=''> | |
| - | You may | + | == Structural highlights == |
| - | + | <table><tr><td colspan='2'>[[1qki]] is a 8 chain structure with sequence from [https://en.wikipedia.org/wiki/Homo_sapiens Homo sapiens]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=1QKI OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=1QKI FirstGlance]. <br> | |
| - | + | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 3Å</td></tr> | |
| - | -- | + | <tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=GOA:GLYCOLIC+ACID'>GOA</scene>, <scene name='pdbligand=GOL:GLYCEROL'>GOL</scene>, <scene name='pdbligand=NAP:NADP+NICOTINAMIDE-ADENINE-DINUCLEOTIDE+PHOSPHATE'>NAP</scene></td></tr> |
| - | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=1qki FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=1qki OCA], [https://pdbe.org/1qki PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=1qki RCSB], [https://www.ebi.ac.uk/pdbsum/1qki PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=1qki ProSAT]</span></td></tr> | |
| + | </table> | ||
| + | == Disease == | ||
| + | [https://www.uniprot.org/uniprot/G6PD_HUMAN G6PD_HUMAN] Defects in G6PD are the cause of chronic non-spherocytic hemolytic anemia (CNSHA) [MIM:[https://omim.org/entry/305900 305900]. Deficiency of G6PD is associated with hemolytic anemia in two different situations. First, in areas in which malaria has been endemic, G6PD-deficiency alleles have reached high frequencies (1% to 50%) and deficient individuals, though essentially asymptomatic in the steady state, have a high risk of acute hemolytic attacks. Secondly, sporadic cases of G6PD deficiency occur at a very low frequencies, and they usually present a more severe phenotype. Several types of CNSHA are recognized. Class-I variants are associated with severe NSHA; class-II have an activity <10% of normal; class-III have an activity of 10% to 60% of normal; class-IV have near normal activity.<ref>PMID:1611091</ref> | ||
| + | == Function == | ||
| + | [https://www.uniprot.org/uniprot/G6PD_HUMAN G6PD_HUMAN] Produces pentose sugars for nucleic acid synthesis and main producer of NADPH reducing power. | ||
| + | == Evolutionary Conservation == | ||
| + | [[Image:Consurf_key_small.gif|200px|right]] | ||
| + | Check<jmol> | ||
| + | <jmolCheckbox> | ||
| + | <scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/qk/1qki_consurf.spt"</scriptWhenChecked> | ||
| + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
| + | <text>to colour the structure by Evolutionary Conservation</text> | ||
| + | </jmolCheckbox> | ||
| + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=1qki ConSurf]. | ||
| + | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
| + | BACKGROUND: Glucose-6-phosphate dehydrogenase (G6PD) catalyses the first committed step in the pentose phosphate pathway; the generation of NADPH by this enzyme is essential for protection against oxidative stress. The human enzyme is in a dimer<-->tetramer equilibrium and its stability is dependent on NADP(+) concentration. G6PD deficiency results from many different point mutations in the X-linked gene encoding G6PD and is the most common human enzymopathy. Severe deficiency causes chronic non-spherocytic haemolytic anaemia; the usual symptoms are neonatal jaundice, favism and haemolytic anaemia. RESULTS: We have determined the first crystal structure of a human G6PD (the mutant Canton, Arg459-->Leu) at 3 A resolution. The tetramer is a dimer of dimers. Despite very similar dimer topology, there are two major differences from G6PD of Leuconostoc mesenteroides: a structural NADP(+) molecule, close to the dimer interface but integral to the subunit, is visible in all subunits of the human enzyme; and an intrasubunit disulphide bond tethers the otherwise disordered N-terminal segment. The few dimer-dimer contacts making the tetramer are charge-charge interactions. CONCLUSIONS: The importance of NADP(+) for stability is explained by the structural NADP(+) site, which is not conserved in prokaryotes. The structure shows that point mutations causing severe deficiency predominate close to the structural NADP(+) and the dimer interface, primarily affecting the stability of the molecule. They also indicate that a stable dimer is essential to retain activity in vivo. As there is an absolute requirement for some G6PD activity, residues essential for coenzyme or substrate binding are rarely modified. | ||
| - | + | Human glucose-6-phosphate dehydrogenase: the crystal structure reveals a structural NADP(+) molecule and provides insights into enzyme deficiency.,Au SW, Gover S, Lam VM, Adams MJ Structure. 2000 Mar 15;8(3):293-303. PMID:10745013<ref>PMID:10745013</ref> | |
| - | + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |
| - | + | </div> | |
| - | + | <div class="pdbe-citations 1qki" style="background-color:#fffaf0;"></div> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | == | + | |
| - | + | ||
==See Also== | ==See Also== | ||
| - | *[[ | + | *[[Glucose 6-phosphate dehydrogenase|Glucose 6-phosphate dehydrogenase]] |
| - | + | == References == | |
| - | == | + | <references/> |
| - | < | + | __TOC__ |
| - | + | </StructureSection> | |
[[Category: Homo sapiens]] | [[Category: Homo sapiens]] | ||
| - | [[Category: | + | [[Category: Large Structures]] |
| - | [[Category: | + | [[Category: Adams MJ]] |
| - | [[Category: | + | [[Category: Au SWN]] |
| - | [[Category: | + | [[Category: Gover S]] |
| - | [[Category: | + | [[Category: Lam VMS]] |
| - | + | ||
| - | + | ||
Current revision
X-RAY STRUCTURE OF HUMAN GLUCOSE 6-PHOSPHATE DEHYDROGENASE (VARIANT CANTON R459L) COMPLEXED WITH STRUCTURAL NADP+
| |||||||||||
Categories: Homo sapiens | Large Structures | Adams MJ | Au SWN | Gover S | Lam VMS