This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
2d1w
From Proteopedia
(Difference between revisions)
m (Protected "2d1w" [edit=sysop:move=sysop]) |
|||
| (9 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | [[Image:2d1w.png|left|200px]] | ||
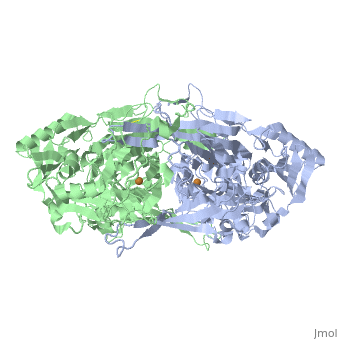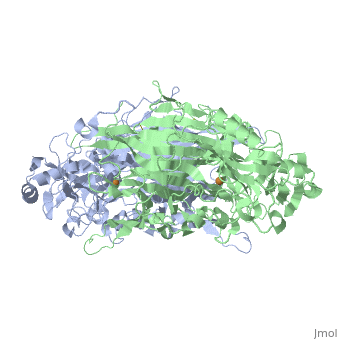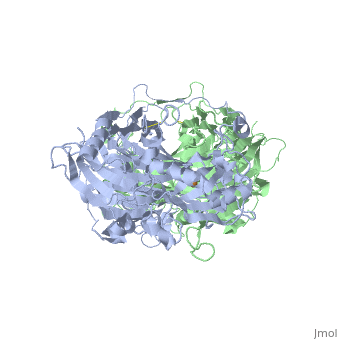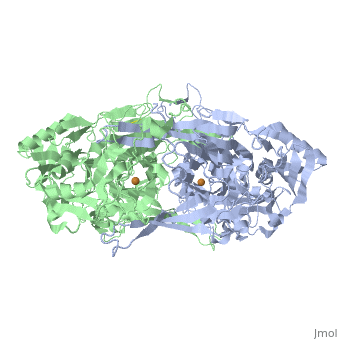
| - | < | + | ==Substrate Schiff-Base intermediate with tyramine in copper amine oxidase from Arthrobacter globiformis== |
| - | + | <StructureSection load='2d1w' size='340' side='right'caption='[[2d1w]], [[Resolution|resolution]] 1.74Å' scene=''> | |
| - | You may | + | == Structural highlights == |
| - | + | <table><tr><td colspan='2'>[[2d1w]] is a 2 chain structure with sequence from [https://en.wikipedia.org/wiki/Arthrobacter_globiformis Arthrobacter globiformis]. Full crystallographic information is available from [http://oca.weizmann.ac.il/oca-bin/ocashort?id=2D1W OCA]. For a <b>guided tour on the structure components</b> use [https://proteopedia.org/fgij/fg.htm?mol=2D1W FirstGlance]. <br> | |
| - | + | </td></tr><tr id='method'><td class="sblockLbl"><b>[[Empirical_models|Method:]]</b></td><td class="sblockDat" id="methodDat">X-ray diffraction, [[Resolution|Resolution]] 1.74Å</td></tr> | |
| - | -- | + | <tr id='ligand'><td class="sblockLbl"><b>[[Ligand|Ligands:]]</b></td><td class="sblockDat" id="ligandDat"><scene name='pdbligand=CU:COPPER+(II)+ION'>CU</scene>, <scene name='pdbligand=TTS:3-((3E)-4-HYDROXY-3-{[2-(4-HYDROXYPHENYL)ETHYL]IMINO}-6-OXOCYCLOHEXA-1,4-DIEN-1-YL)ALANINE'>TTS</scene></td></tr> |
| - | + | <tr id='resources'><td class="sblockLbl"><b>Resources:</b></td><td class="sblockDat"><span class='plainlinks'>[https://proteopedia.org/fgij/fg.htm?mol=2d1w FirstGlance], [http://oca.weizmann.ac.il/oca-bin/ocaids?id=2d1w OCA], [https://pdbe.org/2d1w PDBe], [https://www.rcsb.org/pdb/explore.do?structureId=2d1w RCSB], [https://www.ebi.ac.uk/pdbsum/2d1w PDBsum], [https://prosat.h-its.org/prosat/prosatexe?pdbcode=2d1w ProSAT]</span></td></tr> | |
| + | </table> | ||
| + | == Function == | ||
| + | [https://www.uniprot.org/uniprot/PAOX_ARTGO PAOX_ARTGO] | ||
| + | == Evolutionary Conservation == | ||
| + | [[Image:Consurf_key_small.gif|200px|right]] | ||
| + | Check<jmol> | ||
| + | <jmolCheckbox> | ||
| + | <scriptWhenChecked>; select protein; define ~consurf_to_do selected; consurf_initial_scene = true; script "/wiki/ConSurf/d1/2d1w_consurf.spt"</scriptWhenChecked> | ||
| + | <scriptWhenUnchecked>script /wiki/extensions/Proteopedia/spt/initialview01.spt</scriptWhenUnchecked> | ||
| + | <text>to colour the structure by Evolutionary Conservation</text> | ||
| + | </jmolCheckbox> | ||
| + | </jmol>, as determined by [http://consurfdb.tau.ac.il/ ConSurfDB]. You may read the [[Conservation%2C_Evolutionary|explanation]] of the method and the full data available from [http://bental.tau.ac.il/new_ConSurfDB/main_output.php?pdb_ID=2d1w ConSurf]. | ||
| + | <div style="clear:both"></div> | ||
| + | <div style="background-color:#fffaf0;"> | ||
| + | == Publication Abstract from PubMed == | ||
| + | A key step decisively affecting the catalytic efficiency of copper amine oxidase is stereospecific abstraction of substrate alpha-proton by a conserved Asp residue. We analyzed this step by pre-steady-state kinetics using a bacterial enzyme and stereospecifically deuterium-labeled substrates, 2-phenylethylamine and tyramine. A small and temperature-dependent kinetic isotope effect (KIE) was observed with 2-phenylethylamine, whereas a large and temperature-independent KIE was observed with tyramine in the alpha-proton abstraction step, showing that this step is driven by quantum mechanical hydrogen tunneling rather than the classical transition-state mechanism. Furthermore, an Arrhenius-type preexponential factor ratio approaching a transition-state value was obtained in the reaction of a mutant enzyme lacking the critical Asp. These results provide strong evidence for enzyme-enhanced hydrogen tunneling. X-ray crystallographic structures of the reaction intermediates revealed a small difference in the binding mode of distal parts of substrates, which would modulate hydrogen tunneling proceeding through either active or passive dynamics. | ||
| - | + | Quantum mechanical hydrogen tunneling in bacterial copper amine oxidase reaction.,Murakawa T, Okajima T, Kuroda S, Nakamoto T, Taki M, Yamamoto Y, Hayashi H, Tanizawa K Biochem Biophys Res Commun. 2006 Apr 7;342(2):414-23. Epub 2006 Feb 8. PMID:16487484<ref>PMID:16487484</ref> | |
| - | + | From MEDLINE®/PubMed®, a database of the U.S. National Library of Medicine.<br> | |
| - | + | </div> | |
| - | + | <div class="pdbe-citations 2d1w" style="background-color:#fffaf0;"></div> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | == | + | |
| - | + | ||
==See Also== | ==See Also== | ||
*[[Copper Amine Oxidase|Copper Amine Oxidase]] | *[[Copper Amine Oxidase|Copper Amine Oxidase]] | ||
| - | + | *[[Copper amine oxidase 3D structures|Copper amine oxidase 3D structures]] | |
| - | == | + | == References == |
| - | < | + | <references/> |
| + | __TOC__ | ||
| + | </StructureSection> | ||
[[Category: Arthrobacter globiformis]] | [[Category: Arthrobacter globiformis]] | ||
| - | [[Category: | + | [[Category: Large Structures]] |
| - | [[Category: Hayashi | + | [[Category: Hayashi H]] |
| - | [[Category: Kuroda | + | [[Category: Kuroda S]] |
| - | [[Category: Murakawa | + | [[Category: Murakawa T]] |
| - | [[Category: Nakamoto | + | [[Category: Nakamoto T]] |
| - | [[Category: Okajima | + | [[Category: Okajima T]] |
| - | [[Category: Taki | + | [[Category: Taki M]] |
| - | [[Category: Tanizawa | + | [[Category: Tanizawa K]] |
| - | [[Category: Yamamoto | + | [[Category: Yamamoto Y]] |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
Current revision
Substrate Schiff-Base intermediate with tyramine in copper amine oxidase from Arthrobacter globiformis
| |||||||||||