This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox UC 19
From Proteopedia
(Difference between revisions)
| (3 intermediate revisions not shown.) | |||
| Line 17: | Line 17: | ||
{{Template:ColorKey_Amino2CarboxyRainbow}} | {{Template:ColorKey_Amino2CarboxyRainbow}} | ||
| - | <scene name='Sandbox_UC_19/Leut2/1'>2nd Structure</scene> | ||
| - | {{Template:ColorKey_Helix}} | ||
==Mechanism== | ==Mechanism== | ||
| Line 25: | Line 23: | ||
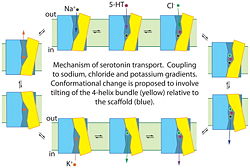
'''Proposed mechanism for serotonin transport by SERT'''. Binding of serotonin (5-HT+), Na+ and Cl− to the transporter from the cell exterior allows a conformational change, shown here as the tilting of a 4-helix bundle (yellow), that closes the extracellular substrate permeation pathway and opens a cytoplasmic pathway. After dissociation of Na+, Cl− and 5-HT+ to the cytoplasm, the transporter binds a K+ ion (or a proton, see (96)) to allow the reverse conformational change, leading to extracellular K+ dissociation and another cycle of transport. | '''Proposed mechanism for serotonin transport by SERT'''. Binding of serotonin (5-HT+), Na+ and Cl− to the transporter from the cell exterior allows a conformational change, shown here as the tilting of a 4-helix bundle (yellow), that closes the extracellular substrate permeation pathway and opens a cytoplasmic pathway. After dissociation of Na+, Cl− and 5-HT+ to the cytoplasm, the transporter binds a K+ ion (or a proton, see (96)) to allow the reverse conformational change, leading to extracellular K+ dissociation and another cycle of transport. | ||
| + | asdasdsdfsadfsdfasfsddf [[transporter|SERT]] sadasdasdasdasd sdfsd wfsdfsd wfsdfsdf | ||
Current revision
Leucine transporter LeuT in complex with sertraline
Recent high-resolution crystal structures of several transporters from protein families that were previously thought to be unrelated show common structural features indicating a large structural family representing transporters from all kingdoms of life.
Content
| |||||||||||