This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Michael Roberts/BIOL115 CaM
From Proteopedia
| (8 intermediate revisions not shown.) | |||
| Line 11: | Line 11: | ||
Click on the ''' 'green links' ''' in the text in the scrollable section below to examine this molecule in more detail. | Click on the ''' 'green links' ''' in the text in the scrollable section below to examine this molecule in more detail. | ||
| - | <StructureSection load='1cll' size=' | + | <StructureSection load='1cll' size='600' side='right' caption='Structure of human calmodulin (PDB entry [[1cll]])' scene='User:Michael_Roberts/BIOL115_CaM/Wireframe/3'> |
== Molecular Model: == | == Molecular Model: == | ||
We'll start with a simple ball-and-stick representation of the protein. This shows all of the atoms that make up the protein and the bonds between them. | We'll start with a simple ball-and-stick representation of the protein. This shows all of the atoms that make up the protein and the bonds between them. | ||
| Line 49: | Line 49: | ||
== Binding to target proteins == | == Binding to target proteins == | ||
| - | '''INACTIVE CALMODULIN:''' | + | '''ACTIVE & INACTIVE CALMODULIN:''' |
| - | At resting levels of cytosolic Ca<sup>2+</sup> (~100 nM), calmodulin exists predominantly in the calcium-free form. This is called apo-calmodulin and <scene name='User:Michael_Roberts/BIOL115_CaM/Inactive_calmodulin/1'> | + | At resting levels of cytosolic Ca<sup>2+</sup> (~100 nM), calmodulin exists predominantly in the calcium-free form. This is called <scene name='User:Michael_Roberts/BIOL115_CaM/Inactive_calmodulin/1'>apo-calmodulin</scene> and its structure is more compact than the structure we saw earlier <scene name='User:Michael_Roberts/BIOL115_CaM/Structure_plus_c/3'>with bound calcium</scene>. Note the extended α-helix linking the two EF-hand-containing domains in the Ca-bound structure, which is interrupted in the <scene name='User:Michael_Roberts/BIOL115_CaM/Inactive_calmodulin/1'>Ca-free form</scene>. Here, the terminal helices are folded down concealing their hydrophobic surfaces and the central chain, which is not now α-helical along its whole length, is not exposed. |
| - | + | ||
| - | + | ||
'''CALMODULIN INTERACTS WITH ITS TARGET:''' | '''CALMODULIN INTERACTS WITH ITS TARGET:''' | ||
| - | The | + | The Ca<sup>2+</sup>-bound form of calmodulin with its exposed hydrophobic surfaces that you have already observed can <scene name='User:Michael_Roberts/BIOL115_CaM/Active_calmodulin/1'>interact with a target protein</scene>. It does this by wrapping around a specific sequence on the target molecule, which is then forced into an α-helical structure. |
The target molecule here (shown in blue) is the calmodulin-regulated enzyme, myosin light chain kinase. Only a short sequence from this protein, the calmodulin binding domain, is shown. | The target molecule here (shown in blue) is the calmodulin-regulated enzyme, myosin light chain kinase. Only a short sequence from this protein, the calmodulin binding domain, is shown. | ||
| + | |||
| + | In this view, <scene name='54/541097/Active_calmodulin/3'>polar and non-polar residues</scene> are coloured in order to highlight the hydrophobic interior of the molecule, which forms the binding site for the myosin light chain kinase calmodulin binding domain. | ||
| + | {{Template:ColorKey_Hydrophobic}}, {{Template:ColorKey_Polar}} | ||
</StructureSection> | </StructureSection> | ||
'''External Resources.''' | '''External Resources.''' | ||
| - | You can view a nice animation of the conformational change undergone by calmodulin upon calcium binding by following this link [http:// | + | You can view a nice animation of the conformational change undergone by calmodulin upon calcium binding by following this link [http://morph2.molmovdb.org/results.rpy?jobid=8350057535]. |
Current revision
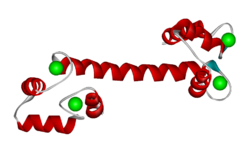
Sequence and structure of EF hands
The EF hand motif is present in a many proteins and it commonly bestows the ability to bind Ca2+ ions. It was first identified in parvalbumin, a muscle protein. Here we'll have a look at the Ca2+-binding protein calmodulin, which possesses four EF hands. Calmodulin and its isoform, troponinC, are important intracellular Ca2+-binding proteins.
The structure below, obtained by X-ray crystallography, represents the Ca2+-binding protein calmodulin. It has a dumbell-shaped structure with two identical lobes connected by a central alpha-helix. Each lobe comprises three α-helices joined by loops. A helix-loop-helix motif forms the basis of each EF hand.
Click on the 'green links' in the text in the scrollable section below to examine this molecule in more detail.
| |||||||||||
External Resources. You can view a nice animation of the conformational change undergone by calmodulin upon calcium binding by following this link [1].
