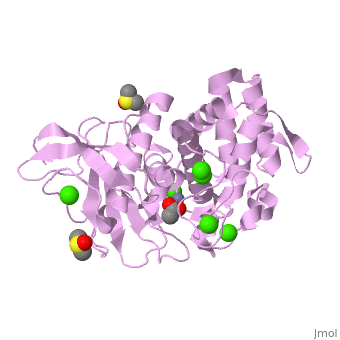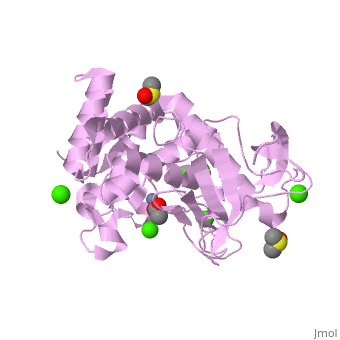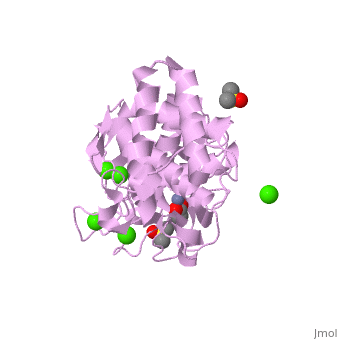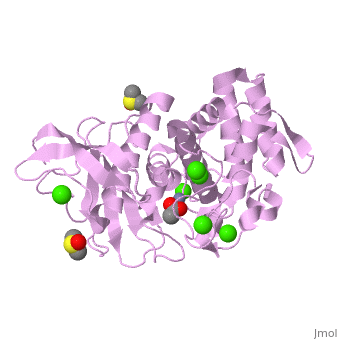This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Thermolysin
From Proteopedia
(Difference between revisions)
| (15 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | <StructureSection load='2a7g' size=' | + | <StructureSection load='2a7g' size='350' side='right' scene= caption='Thermolysin complex with acetate, DMS, Zn+2 (grey) and Ca+2 (green) ions, [[2a7g]]'> |
| + | == Function == | ||
| + | [[Thermolysin]] or '''thermostable neutral proteinase''' (TML) is a thermostable metalloproteinase enzyme from ''Bacillus thermoproteolyticus''. It catalyzes the hydrolysis of peptide bonds containing hydrophobic residues. See [[Metalloproteases]] and [[Matrix metalloproteinase]] for discussion. | ||
| - | + | == Structural highlights == | |
| - | + | Thermolysin is a well researched [[metalloproteases|metalloprotease]] containing <scene name='User:Ralf_Stephan/Sandbox_2/Zinc/2'>zinc</scene> (click this!) and the amino acids His-Glu-X-His-His as its catalytic center. <scene name='User:Ralf_Stephan/Sandbox_2/Res_yellow/3'>Glu-166, His-142 and -146 are grouped around the zinc atom</scene>, holding it fast, while <scene name='User:Ralf_Stephan/Sandbox_2/Res/1'>Glu-143 holds the polarized water atom. Additionally, Tyr-157 and His-231</scene> stabilize the substrate protein which will be cleaved into two smaller proteins.<ref>Matthews, BW. (1988): ''Structural basis of the action of thermolysin and related zinc peptidases''. In: ''Acc. Chem. Res.'' '''21'''(9); 333–340; http://dx.doi.org/10.1021/ar00153a003</ref><ref>PMID:11935352</ref>. | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
== 3D Structures of Thermolysin == | == 3D Structures of Thermolysin == | ||
| - | + | [[Thermolysin 3D structures]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | [[ | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
</StructureSection> | </StructureSection> | ||
| - | ==References== | + | == References == |
| - | <references /> | + | <references/> |
| - | + | [[Category: Topic Page]] | |
| - | [[Category:Topic Page]] | + | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
Current revision
| |||||||||||
References
- ↑ Matthews, BW. (1988): Structural basis of the action of thermolysin and related zinc peptidases. In: Acc. Chem. Res. 21(9); 333–340; http://dx.doi.org/10.1021/ar00153a003
- ↑ Pelmenschikov V, Blomberg MR, Siegbahn PE. A theoretical study of the mechanism for peptide hydrolysis by thermolysin. J Biol Inorg Chem. 2002 Mar;7(3):284-98. Epub 2001 Sep 27. PMID:11935352 doi:http://dx.doi.org/10.1007/s007750100295