This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Sandbox 124
From Proteopedia
(Difference between revisions)
| (16 intermediate revisions not shown.) | |||
| Line 3: | Line 3: | ||
==='''Introduction'''=== | ==='''Introduction'''=== | ||
| - | + | Peptidoglycan transpeptidases, also known as penicillin-binding proteins | |
| - | , also known as penicillin-binding proteins | + | |
(PBP), catalyze the cross-linking of peptidoglycan polymers during | (PBP), catalyze the cross-linking of peptidoglycan polymers during | ||
bacterial cell wall synthesis. Beta-lactam (β-lactam) antibiotics, | bacterial cell wall synthesis. Beta-lactam (β-lactam) antibiotics, | ||
| Line 15: | Line 14: | ||
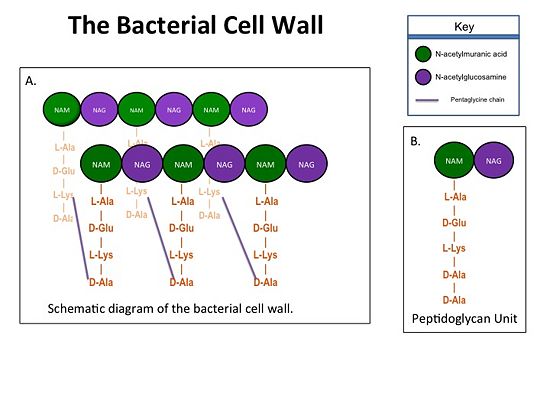
| - | '''Cell Wall Structure''' | + | ==='''Cell Wall Structure'''=== |
The cell wall, which is composed of peptidoglycans, is crucial for maintaining | The cell wall, which is composed of peptidoglycans, is crucial for maintaining | ||
the structural integrity of the bacterium. Peptidoglycans consists of | the structural integrity of the bacterium. Peptidoglycans consists of | ||
| Line 30: | Line 29: | ||
==='''Catalytic Mechanism of PBP2a'''=== | ==='''Catalytic Mechanism of PBP2a'''=== | ||
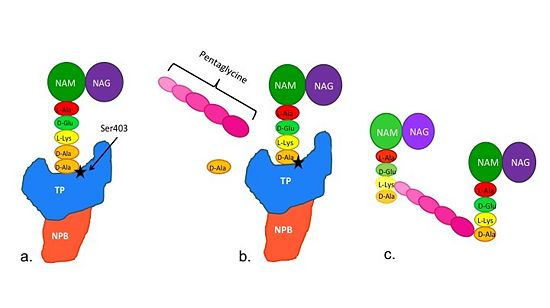
| - | [[Image:Schematic TP 3steps.jpg|thumb|alt= Alt text| |550px]] | + | [[Image:Schematic TP 3steps.jpg|thumb|alt= Alt text|Figure 2. Schematic showing Catalytic Mechanism of PBP2a |550px]] |
(a) The D-Ala-D-Ala side-chain substrate of the peptidoglycan accesses | (a) The D-Ala-D-Ala side-chain substrate of the peptidoglycan accesses | ||
the active site of the PBP2a. | the active site of the PBP2a. | ||
| Line 44: | Line 43: | ||
The entire process takes 4 milliseconds. | The entire process takes 4 milliseconds. | ||
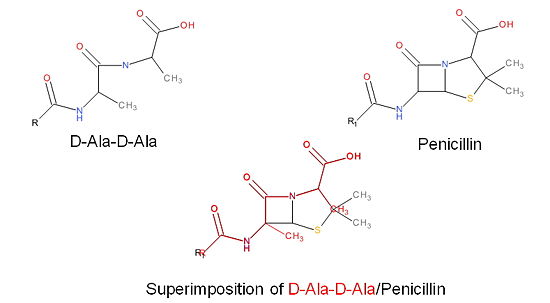
| - | ==='''How | + | ==='''How Do Antibiotics Work?'''=== |
The β-lactam antibiotics inhibit bacterial growth by inhibiting PBPs and ultimately cell wall | The β-lactam antibiotics inhibit bacterial growth by inhibiting PBPs and ultimately cell wall | ||
synthesis. Specifically, β-lactams are molecular mimics of D-Ala-D-Ala, which is the normal | synthesis. Specifically, β-lactams are molecular mimics of D-Ala-D-Ala, which is the normal | ||
| Line 50: | Line 49: | ||
inhibited by the β-lactam. As a result, the synthesis of the cell wall is inhibited which leads | inhibited by the β-lactam. As a result, the synthesis of the cell wall is inhibited which leads | ||
to cell lysis. | to cell lysis. | ||
| + | [[Image:Structures on penicillin and b lactam.jpg|thumb|alt= Alt text|Figure 3. Mechanism of action of β-lactams. A. Structure of a β-lactam (penicillin) showing the amide, carboxyl, and β-lactam ring groups β-lactam ring groups. B. Structure of the D-Ala-D-Ala substrate. C. Overlay of the D-Ala-D-Ala substrate in red with penicillin demonstrating molecular mimicry.|550 px]] | ||
| + | |||
==='''PBP2a and Ceftobiprole'''=== | ==='''PBP2a and Ceftobiprole'''=== | ||
| Line 56: | Line 57: | ||
<scene name='37/372724/Ceftobiprole/1'>ceftobiprole</scene> and | <scene name='37/372724/Ceftobiprole/1'>ceftobiprole</scene> and | ||
ceftaroline – that have anti-MRSA activity have been developed. Ceftobiprole is able to | ceftaroline – that have anti-MRSA activity have been developed. Ceftobiprole is able to | ||
| - | inhibit PBP2a because additional chemical groups at the <scene name='37/372724/ | + | inhibit PBP2a because additional chemical groups at the |
| + | <scene name='37/372724/Ceftobiprole/7'>R2</scene> | ||
position of the cephalosporin backbone are able to interact with additional amino acid | position of the cephalosporin backbone are able to interact with additional amino acid | ||
| - | residues in PBP2a; specifically <scene name='37/372724/ | + | residues in PBP2a; specifically |
| - | As a result of | + | <scene name='37/372724/Tyr446_and_met641_label/2'>Tyr446 and Met641</scene>. |
| - | + | As a result of ceftobiprole <scene name='37/372724/R2_interaction/4'>tighter binding</scene> to PBP2a as highlighted in green , <scene name='37/372724/Ceftobiprole_in_cpk/1'>the medicine</scene>, shown as colors of the atom types ([[CPK]]), is able to more efficiently react with the serine active site residue and therefore inhibit the activity of PBP2a. | |
Current revision
| |||||||||||