This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Exonuclease
From Proteopedia
(Difference between revisions)
| (15 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
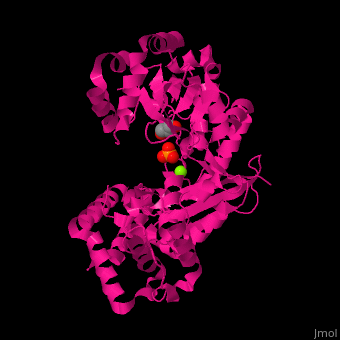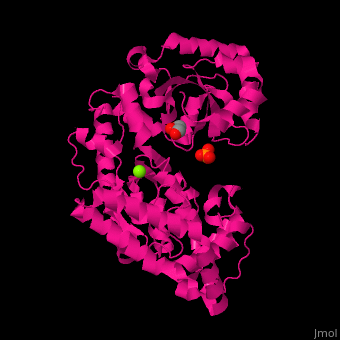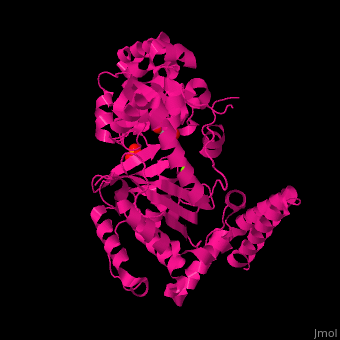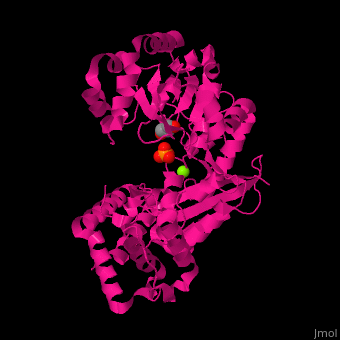
| - | + | <StructureSection load='' size='350' side='right' scene='46/466467/Cv/1' caption='E. coli exonuclease I complex with phosphate, glycerol and Mg+2 ion (PDB code [[1fxx]]) '> | |
| - | '''Exonucleases''' (ExN) cleave nucleotides at the end of a polynucleotide chain. In E. coli there are ExN I to VIII.<br /> | + | |
| + | '''Exonucleases''' (ExN) cleave nucleotides at the end of a polynucleotide chain<ref>PMID:14770007</ref>. In ''E. coli'' there are ExN I to VIII. ExN were found in human and yeast as well. | ||
| + | <br /> | ||
* '''ExN-I''' cleaves single-stranded DNA.<br /> | * '''ExN-I''' cleaves single-stranded DNA.<br /> | ||
* '''ExN-II''' is associated with DNA polymerase.<br /> | * '''ExN-II''' is associated with DNA polymerase.<br /> | ||
| Line 8: | Line 10: | ||
* '''ExN-V''' cleaves linear double-stranded DNA.<br /> | * '''ExN-V''' cleaves linear double-stranded DNA.<br /> | ||
* '''ExN-VIII''' requires a free 5’ OH group for reacting.<br /> | * '''ExN-VIII''' requires a free 5’ OH group for reacting.<br /> | ||
| - | * '''ExN''' | + | * For '''phage ExN''' see [[Sand box 211]]. |
| + | |||
| + | <scene name='46/466467/Cv/3'>Mg coordination site</scene> in ''E. coli'' exonuclease I (PDB code [[1fxx]]).<ref>PMID:11101894</ref> Water molecules shown as red spheres. | ||
== 3D Structures of exonuclease == | == 3D Structures of exonuclease == | ||
| + | [[Exonuclease 3D structures]] | ||
| - | + | </StructureSection> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| + | == References == | ||
| + | <references/> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
References
- ↑ Mukherjee D, Fritz DT, Kilpatrick WJ, Gao M, Wilusz J. Analysis of RNA exonucleolytic activities in cellular extracts. Methods Mol Biol. 2004;257:193-212. PMID:14770007 doi:http://dx.doi.org/10.1385/1-59259-750-5:193
- ↑ Breyer WA, Matthews BW. Structure of Escherichia coli exonuclease I suggests how processivity is achieved. Nat Struct Biol. 2000 Dec;7(12):1125-8. PMID:11101894 doi:10.1038/81978