This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Farnesyltransferase
From Proteopedia
(Difference between revisions)
| (15 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
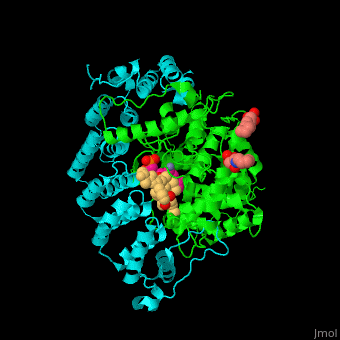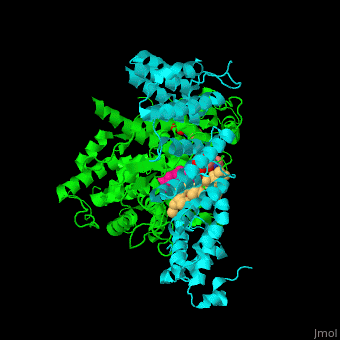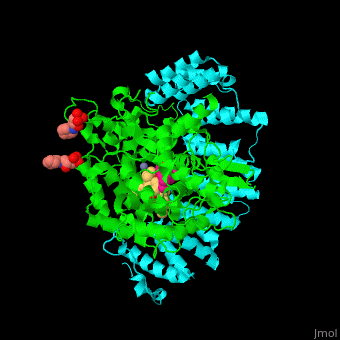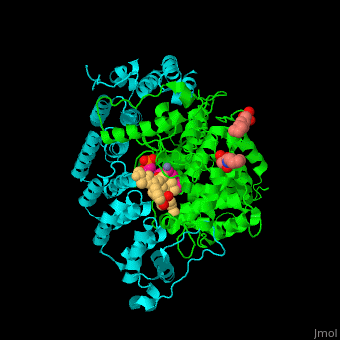
| - | + | <StructureSection load='' size='350' side='right' scene='48/485616/Cv/2' caption=' Farnesyltransferase α subunit (cyan) and β subunit (green) complex with farnesyl diphosphate (FPP) analog, Zn+2 ion (grey), sulfonic acid derivative and CAAX peptide (PDB code [[3q75]]) '> | |
| + | == Function == | ||
| - | + | '''Farnesyltransferase''' (FTase) is part of the prenyltransferase group. FTase modifies proteins by adding farnesyl diphosphate (FPP) – an isoprenoid lipid – to a cysteine in a CAAX motif near the C terminal. This addition forms a thioether linkage, makes the protein more hydrophobic and associates it with the membrane. Farnesylated proteins – like those of the Ras family - are involved in cellular signaling<ref>PMID:15451670</ref>. | |
| - | + | ||
| - | + | == Relevance == | |
| - | + | ||
| - | + | ||
| - | '''Farnesyltransferase''' (FTase) is part of the prenyltransferase group. FTase modifies proteins by adding farnesyl diphosphate (FPP) – an isoprenoid lipid – to a cysteine near the C terminal. This addition forms a thioether linkage, makes the protein more hydrophobic and associates it with the membrane. Farnesylated proteins – like those of the Ras family - are involved in cellular signaling. | + | |
| - | + | FTase inhibitors are being tested as anti-cancer and anti-Progeria agents. | |
| - | + | == Structural insights == | |
| - | + | ||
| - | + | FTase are composed of 2 subunits. <scene name='48/485616/Cv/11'>α subunit</scene> is in cyan, <scene name='48/485616/Cv/12'>β subunit</scene> is in green. <scene name='48/485616/Cv/13'>CAAX peptide and ligands</scene>. The <scene name='48/485616/Cv/14'>CAAX motif and β subunit coordinate with the Zn+2 ion</scene> and the <scene name='48/485616/Cv/15'>FPP predominantly coordinates with the β subunit</scene><ref>PMID:21816822</ref>. Water molecules are shown as red spheres. | |
| - | + | ==3D structures of farnesyltransferase== | |
| - | + | [[Farnesyltransferase 3D structures]] | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | </StructureSection> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | == References == | |
| + | <references/> | ||
| - | **[[1n4p]], [[1n4r]], [[1n4s]] - rFTase α subunit + GTase β subunit + transforming protein + GPP<br /> | ||
| - | }} | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
References
- ↑ Reid TS, Terry KL, Casey PJ, Beese LS. Crystallographic analysis of CaaX prenyltransferases complexed with substrates defines rules of protein substrate selectivity. J Mol Biol. 2004 Oct 15;343(2):417-33. PMID:15451670 doi:10.1016/j.jmb.2004.08.056
- ↑ Hast MA, Nichols CB, Armstrong SM, Kelly SM, Hellinga HW, Alspaugh JA, Beese LS. Structures of Cryptococcus neoformans Protein Farnesyltransferase Reveal Strategies for Developing Inhibitors That Target Fungal Pathogens. J Biol Chem. 2011 Oct 7;286(40):35149-62. Epub 2011 Aug 4. PMID:21816822 doi:10.1074/jbc.M111.250506