This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Journal:Cell:1
From Proteopedia
(Difference between revisions)

| (2 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
| - | + | <StructureSection load='' size='400' side='right' scene='User:David_Canner/Workbench/Opening/2' caption='Solved Structures of IFNAR1/2/IFN Complexes, [[1dq8]]'> | |
=== Structural linkage between ligand discrimination and receptor activation by type I interferons === | === Structural linkage between ligand discrimination and receptor activation by type I interferons === | ||
<big>Christoph Thomas, Ignacio Moraga, Doron Levin, Peter O. Krutzik, Yulia Podoplelova, Angelica Trejo, Choongho Lee, Ganit Yarden, Susan E. Vleck, Jeffrey S. Glenn, Garry P. Nolan, Jacob Piehler, Gideon Schreiber, K. Christopher Garcia</big><ref>DOI 10.1016/j.cell.2011.06.048</ref> | <big>Christoph Thomas, Ignacio Moraga, Doron Levin, Peter O. Krutzik, Yulia Podoplelova, Angelica Trejo, Choongho Lee, Ganit Yarden, Susan E. Vleck, Jeffrey S. Glenn, Garry P. Nolan, Jacob Piehler, Gideon Schreiber, K. Christopher Garcia</big><ref>DOI 10.1016/j.cell.2011.06.048</ref> | ||
| Line 36: | Line 36: | ||
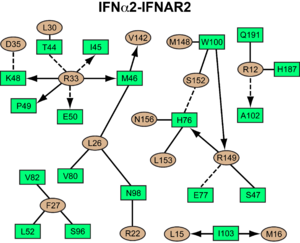
The low-affinity receptor chain, IFNAR1, also <scene name='User:David_Canner/Workbench3/Morph_4/4'>undergoes major conformational changes</scene> upon ligand binding. When using D1 as anchor, D3 is moving inwards (toward the ligand) by ~15 Å. This would generate an even larger movement of the membrane-proximal SD4 domain and the transmembrane helix. The conformational changes in IFNAR1 are necessary to form the full spectrum of interactions with the IFN ligand, and to form a stable signaling complex that is able to instigate downstream signaling. In contrast to SD3, SD4 seems to be highly flexible (even more than D2 of IFNAR2). One might suggest that the conformational changes in IFNAR1 by itself will be responsible for a reduced binding affinity of IFNAR1 and may slow down the rate of ligand association to IFNAR1 directly from solution. | The low-affinity receptor chain, IFNAR1, also <scene name='User:David_Canner/Workbench3/Morph_4/4'>undergoes major conformational changes</scene> upon ligand binding. When using D1 as anchor, D3 is moving inwards (toward the ligand) by ~15 Å. This would generate an even larger movement of the membrane-proximal SD4 domain and the transmembrane helix. The conformational changes in IFNAR1 are necessary to form the full spectrum of interactions with the IFN ligand, and to form a stable signaling complex that is able to instigate downstream signaling. In contrast to SD3, SD4 seems to be highly flexible (even more than D2 of IFNAR2). One might suggest that the conformational changes in IFNAR1 by itself will be responsible for a reduced binding affinity of IFNAR1 and may slow down the rate of ligand association to IFNAR1 directly from solution. | ||
| + | |||
| + | Coordinates and structure factors have been deposited within the Research Collaboratory for Structural Bioinformatics (RCSB) Protein Data Bank (PDB) under accession codes [[3s98]] (IFNAR1ΔSD4), [[3s8w]] (IFNAR2-D2), [[3s9d]] (binary IFNα-IFNAR2 complex), [[3se4]] (ternary IFNω complex), and [[3se3]] (ternary IFNα complex). | ||
__NOTOC__ | __NOTOC__ | ||
</StructureSection> | </StructureSection> | ||
Current revision
| |||||||||||
- ↑ Thomas C, Moraga I, Levin D, Krutzik PO, Podoplelova Y, Trejo A, Lee C, Yarden G, Vleck SE, Glenn JS, Nolan GP, Piehler J, Schreiber G, Garcia KC. Structural Linkage between Ligand Discrimination and Receptor Activation by Type I Interferons. Cell. 2011 Aug 19;146(4):621-32. PMID:21854986 doi:10.1016/j.cell.2011.06.048
Proteopedia Page Contributors and Editors (what is this?)
Christoph Thomas, Jaime Prilusky, Joel L. Sussman, Michal Harel, Alexander Berchansky
This page complements a publication in scientific journals and is one of the Proteopedia's Interactive 3D Complement pages. For aditional details please see I3DC.