This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Renin
From Proteopedia
(Difference between revisions)
| (6 intermediate revisions not shown.) | |||
| Line 2: | Line 2: | ||
{{Clear}} | {{Clear}} | ||
| - | '''Renin''', also known as angiotensinogenase, is an aspartyl protease and belongs to the protein family peptidase A1. Aspartyl proteases are endopeptidases that typically use two aspartate residues in the active site to specifically cleave peptide substrates using an acid-base hydrolysis mechanism. Mature renin circulates in the blood stream and contains 340 amino acid residues and has a mass of approximately 37 kDa. The function of renin is to cleave angiotensinogen to produce angiotensin I. | + | == Introduction == |
| + | '''Renin''', also known as '''angiotensinogenase''', is an aspartyl protease and belongs to the protein family peptidase A1. Aspartyl proteases are endopeptidases that typically use two aspartate residues in the active site to specifically cleave peptide substrates using an acid-base hydrolysis mechanism. Mature renin circulates in the blood stream and contains 340 amino acid residues and has a mass of approximately 37 kDa. The function of renin is to cleave angiotensinogen to produce angiotensin I. | ||
Renin is secreted by the kidneys. The kidneys act both directly and indirectly to regulate arterial blood pressure and provide the major long term mechanism of blood pressure and control. The direct mechanism changes blood volume independently of hormones. When blood pressure and blood volume increase the kidneys can not filter all of the liquids and thus liquids are lost in the urine to decrease blood pressure and blood volume. | Renin is secreted by the kidneys. The kidneys act both directly and indirectly to regulate arterial blood pressure and provide the major long term mechanism of blood pressure and control. The direct mechanism changes blood volume independently of hormones. When blood pressure and blood volume increase the kidneys can not filter all of the liquids and thus liquids are lost in the urine to decrease blood pressure and blood volume. | ||
| Line 38: | Line 39: | ||
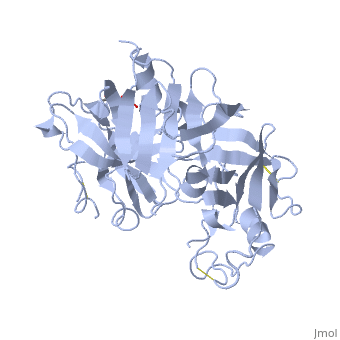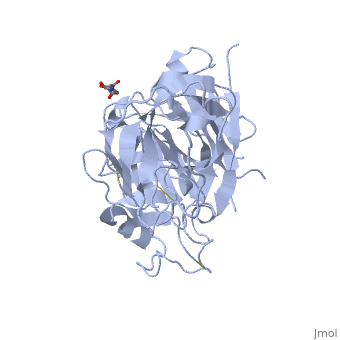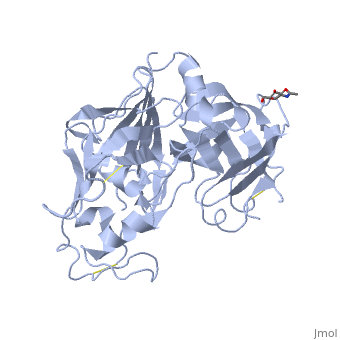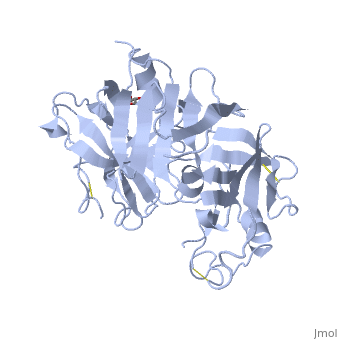
[[Image:Aliskiren.jpg|thumb|400px|alt=text|Aliskiren<ref>PMID:15723979</ref>]] | [[Image:Aliskiren.jpg|thumb|400px|alt=text|Aliskiren<ref>PMID:15723979</ref>]] | ||
{{Clear}} | {{Clear}} | ||
| - | <scene name=' | + | <scene name='10/1020133/Cv/3'>Renin with bound Aliskiren</scene> ([[2v0z]]). |
| - | + | ||
| - | Aliskiren is a hydrophilic molecule. When bound to renin, aliskiren occupies the S1, S1', S2', and S3 hydrophobic regions of renin. | + | <scene name='10/1020133/Cv/9'>Aliskiren binding site</scene>. Water molecules are shown as red spheres. |
| + | |||
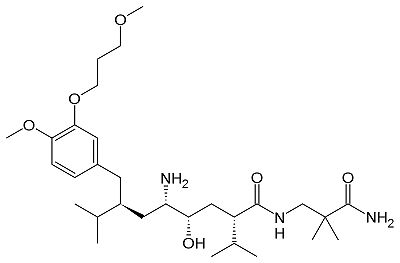
| + | [[Aliskiren]] is a hydrophilic molecule. When bound to renin, aliskiren occupies the S1, S1', S2', and S3 hydrophobic regions of renin. Most importantly aliskiren occupies the S3<sup>SP</sup> region that is equally hydrophobic and hydrophilic and greatly increases binding affinity.<ref>PMID: 20731374</ref> Aliskiren interacts with multiple residues in renin. The hydroxyl group hydrogen bonds both <scene name='10/1020133/Asp32/1'>aspartate 32</scene> oxygens. The methoxy group in the S3 hydrophobic region hydrogen bonds to secondary amine group of <scene name='10/1020133/Tyr14/1'>tyrosine 14</scene>. The amide group hydrogen bonds with the secondary amine of <scene name='10/1020133/Ser76/1'>serine 76</scene>.<ref>PMID: 20855222</ref> And the terminal amide hydrogen bonds with <scene name='10/1020133/Arg74/1'>arginine 74</scene> in the S2' hydrophobic pocket.<ref>PMID: 21708467</ref> | ||
| + | |||
| + | There are three generations of renin inhibitors. The first two generation molecules were peptide molecules. These peptide molecules were not specific or effective as renin inhibitors. Aliskiren, part of the 3<sup>rd</sup> generation, is a nonpeptide renin inhibitor. Small molecule nonpeptide inhibitors such as aliskiren have good pharmokenetics and are very specific for renin and not other protein peptidases. Advancements in crystallography and molecular modeling allowed the discovery of aliskiren. Aliskiren inhibits renin activity. Since renin is the rate limiting step of the RAS renin inhibition is a successful method to lower blood pressure. | ||
Aliskiren is approved by the [http://www.fda.gov/ Federal Drug Administration (FDA)] to treat hypertension. | Aliskiren is approved by the [http://www.fda.gov/ Federal Drug Administration (FDA)] to treat hypertension. | ||
| Line 54: | Line 58: | ||
Familial juvenile hyperuricemic nephropathy type 2 is also caused by defects in the renin gene. Familial juvenile hyperuricemic nephropathy type 2 is characterized by slowly progressive renal failure and anemia. The autosomal dominant disorder is caused by a deletion of leucine 16 or a mutation of leucine 16 to arginine. The mutations effect the hydrophobicity of the signal sequence and disrupt the proper transport of preprorenin into the endoplasmic reticulum and thus effecting prerenin processing. The mutatnt proteins are toxic and reduce the viability of renin expressing cells, eventually causing renal failure.<ref>PMID:19664745</ref> | Familial juvenile hyperuricemic nephropathy type 2 is also caused by defects in the renin gene. Familial juvenile hyperuricemic nephropathy type 2 is characterized by slowly progressive renal failure and anemia. The autosomal dominant disorder is caused by a deletion of leucine 16 or a mutation of leucine 16 to arginine. The mutations effect the hydrophobicity of the signal sequence and disrupt the proper transport of preprorenin into the endoplasmic reticulum and thus effecting prerenin processing. The mutatnt proteins are toxic and reduce the viability of renin expressing cells, eventually causing renal failure.<ref>PMID:19664745</ref> | ||
| - | </StructureSection> | ||
| - | __NOTOC__ | ||
==3D structures of renin== | ==3D structures of renin== | ||
| Line 63: | Line 65: | ||
[[1bbs]], [[2ren]] – hRen – human<br /> | [[1bbs]], [[2ren]] – hRen – human<br /> | ||
[[1rne]] – hRen + transition state analog inhibitor <br /> | [[1rne]] – hRen + transition state analog inhibitor <br /> | ||
| - | [[1hrn]], [[1bil]], [[1bim]], [[2bks]], [[2bkt]], [[2fs4]], [[2g1n]], [[2g1o]], [[2g1r]], [[2g1s]], [[2g1y]], [[2g21]], [[2g22]], [[2g24]], [[2g26]], [[2g27]], [[2g20]], [[2i4q]], [[2iko]], [[2iku]], [[2il2]], [[2v0z]], [[2v10]], [[2v11]], [[3d91]], [[2v13]], [[2v16]], [[3gw5]], [[3g6z]], [[3g70]], [[3g72]], [[3km4]], [[3k1w]], [[3oqf]], [[3oot]], [[3oqk]], [[3oad]], [[3oag]], [[3own]], [[3o9l]], [[3q3t]], [[3sfc]], [[3q4b]], [[3q5h]], [[3vsw]], [[3vsx]], [[2v12]], [[3vuc]], [[3vyd]], [[3vye]], [[3vyf]], [[4gj5]], [[4gj6]], [[4gj7]], [[4gj8]], [[4gj9]], [[4gja]], [[4gjb]], [[4gjc]], [[4gjd]], [[4pyv]], [[4q1n]], [[4ryc]], [[4ryg]], [[4rz1]], [[4s1g]], [[4xx3]], [[4xx4]] - hRen + inhibitor<br /> | + | [[1hrn]], [[1bil]], [[1bim]], [[2bks]], [[2bkt]], [[2fs4]], [[2g1n]], [[2g1o]], [[2g1r]], [[2g1s]], [[2g1y]], [[2g21]], [[2g22]], [[2g24]], [[2g26]], [[2g27]], [[2g20]], [[2i4q]], [[2iko]], [[2iku]], [[2il2]], [[2v0z]], [[2v10]], [[2v11]], [[3d91]], [[2v13]], [[2v16]], [[3gw5]], [[3g6z]], [[3g70]], [[3g72]], [[3km4]], [[3k1w]], [[3oqf]], [[3oot]], [[3oqk]], [[3oad]], [[3oag]], [[3own]], [[3o9l]], [[3q3t]], [[3sfc]], [[3q4b]], [[3q5h]], [[3vsw]], [[3vsx]], [[2v12]], [[3vuc]], [[3vyd]], [[3vye]], [[3vyf]], [[4gj5]], [[4gj6]], [[4gj7]], [[4gj8]], [[4gj9]], [[4gja]], [[4gjb]], [[4gjc]], [[4gjd]], [[4pyv]], [[4q1n]], [[4ryc]], [[4ryg]], [[4rz1]], [[4s1g]], [[4xx3]], [[4xx4]], [[5kod]], [[5kos]], [[5kot]], [[5t4s]], [[5sxn]], [[5sy2]], [[5sy3]], [[5sz9]], [[5koq]], [[5tmg]], [[5tmk]], [[5v8v]], [[5vpm]], [[5vrp]], [[7xgk]], [[7xgo]], [[7xgp]] - hRen + inhibitor<br /> |
[[2x0b]] – hRen + angiotensinogen<br /> | [[2x0b]] – hRen + angiotensinogen<br /> | ||
| - | [[1smr]] - | + | [[6i3f]] – hRen (mutant) + angiotensinogen<br /> |
| + | [[5mkt]] – mRen-1 – mouse<br /> | ||
| + | [[1smr]] - mRen + peptide inhibitor<br /> | ||
| + | [[5mlg]] – Ren – rat<br /> | ||
[[3vcm]], [[4amt]] – hProrenin | [[3vcm]], [[4amt]] – hProrenin | ||
| Line 71: | Line 76: | ||
==References== | ==References== | ||
<references /> | <references /> | ||
| + | </StructureSection> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||