This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
User:Scot Wherland/Sandbox 1
From Proteopedia
< User:Scot Wherland(Difference between revisions)
| (5 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
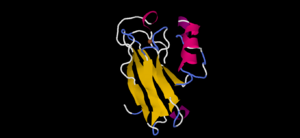
| - | [[Image:4AZUa.png|300px|left|thumb| Ps. aer Azurin]] <Structure load='4AZU' size='400' frame='true' align='right' caption='Ps. aer Azurin' scene='Insert optional scene name here' /> | + | [[Image:4AZUa.png|300px|left|thumb| Ps. aer Azurin]] |
| + | <StructureSection> | ||
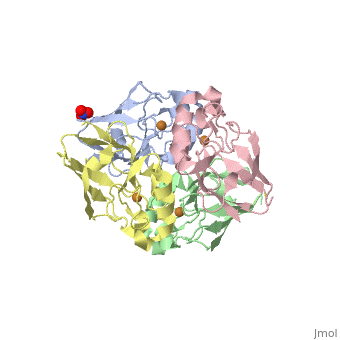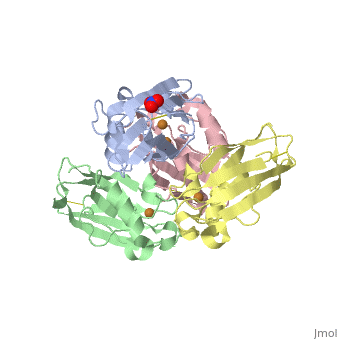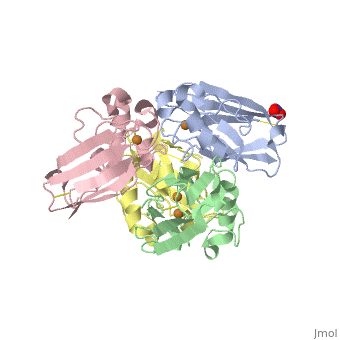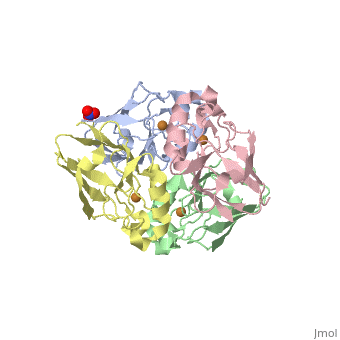
| + | <Structure load='4AZU' size='400' frame='true' align='right' caption='Ps. aer Azurin' scene='Insert optional scene name here' /> | ||
| + | |||
== Introduction == | == Introduction == | ||
| Line 19: | Line 22: | ||
Electron transfer is slowed by distance between the donor and acceptor, but this can be partially ameliorated by an appropriately constructed pathway of covalent bonds. Thus the distance between the electron donor and acceptor can be analyzed in terms of pathways involving covalent bonds, hydrogen bonds (less effective), and through-space jumps (even less effective). One pathway between the disulfide and copper involves a covalent path from SG of Cys 3, through the backbone Ser 4, Val 5, Asp 6, Ile 7, Gln 8, Gly 9 and Asn 10, then through a hydrogen bond from the O of Asn 10 to the proton on NE2 (Ne) of the ligand His46. The second path branches through a hydrogen bond from the carbonyl O of Cys 3 to the peptide N of Thr 30, then through the backbone of Val 31 and then via a through-space jump from the side chain CG of Val 31 to CG of the sidechain of Trp 48, then through the side chain and backbone of Trp 48 and Val 49, followed by a hydrogen bond from the backbone N of Val 49 to to carbonyl O of Phe 111 and then to the Cu via the ligand Cys 112. The orbital coupling provide by this path is sensitive to the distance of the through-space jump, and thus is influenced by the mobility of the structure. <scene name='User:Scot_Wherland/Sandbox_1/Azurinpath1/1'>Path 1</scene> <scene name='User:Scot_Wherland/Sandbox_1/Azurinpath2/5'>Path 2</scene> | Electron transfer is slowed by distance between the donor and acceptor, but this can be partially ameliorated by an appropriately constructed pathway of covalent bonds. Thus the distance between the electron donor and acceptor can be analyzed in terms of pathways involving covalent bonds, hydrogen bonds (less effective), and through-space jumps (even less effective). One pathway between the disulfide and copper involves a covalent path from SG of Cys 3, through the backbone Ser 4, Val 5, Asp 6, Ile 7, Gln 8, Gly 9 and Asn 10, then through a hydrogen bond from the O of Asn 10 to the proton on NE2 (Ne) of the ligand His46. The second path branches through a hydrogen bond from the carbonyl O of Cys 3 to the peptide N of Thr 30, then through the backbone of Val 31 and then via a through-space jump from the side chain CG of Val 31 to CG of the sidechain of Trp 48, then through the side chain and backbone of Trp 48 and Val 49, followed by a hydrogen bond from the backbone N of Val 49 to to carbonyl O of Phe 111 and then to the Cu via the ligand Cys 112. The orbital coupling provide by this path is sensitive to the distance of the through-space jump, and thus is influenced by the mobility of the structure. <scene name='User:Scot_Wherland/Sandbox_1/Azurinpath1/1'>Path 1</scene> <scene name='User:Scot_Wherland/Sandbox_1/Azurinpath2/5'>Path 2</scene> | ||
| - | + | </StructureSection> | |
== See Also == | == See Also == | ||
== References == | == References == | ||
Current revision
| |||||||||||