This old version of Proteopedia is provided for student assignments while the new version is undergoing repairs. Content and edits done in this old version of Proteopedia after March 1, 2026 will eventually be lost when it is retired in about June of 2026.
Apply for new accounts at the new Proteopedia. Your logins will work in both the old and new versions.
Acyl carrier protein
From Proteopedia
(Difference between revisions)
| (23 intermediate revisions not shown.) | |||
| Line 1: | Line 1: | ||
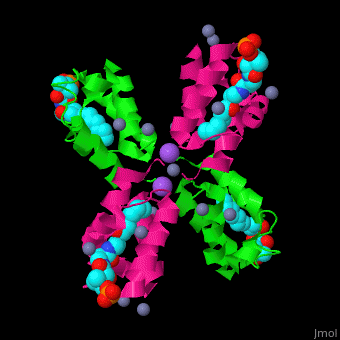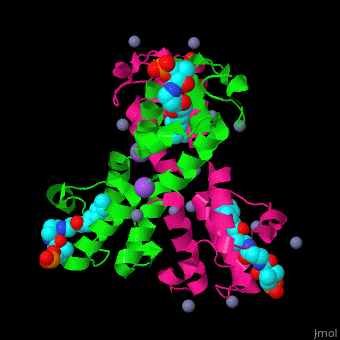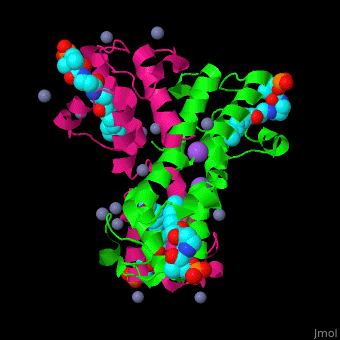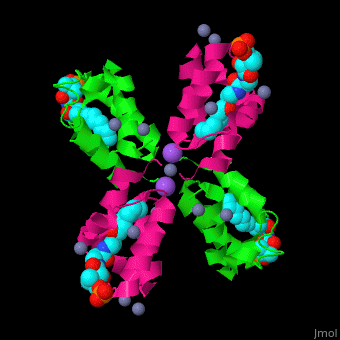
| - | <StructureSection load=' | + | <StructureSection load='' size='350' side='right' caption='Structure of E. coli acyl carrier protein complex with thioester, Zn+2 (grey) and Na+ (purple) ions (PDB code [[2fad]]).' scene='59/592131/Cv/1'> |
== Function == | == Function == | ||
'''Acyl carrier protein''' (ACP) is a component of the fatty acid biosynthesis cycle. ACP catalyzes the addition of a thioester to a phosphopantetheine moiety. The phosphopantetheine moiety is added post-translationally to ACP serine residue by ACP synthetase (ACPS). The phosphopantetheine contains a free SH group which binds acyl groups in the fatty acid biosynthesis as thioesters. The acyl groups from acetyl-CoA and malonyl-CoA are transferred to ACP. There are 2 types of ACP. '''ACP I''' is a multifunctional polypeptide found in yeast and mammals while '''ACP II''' is a monomeric protein found in bacteria and plants.<ref>PMID:18059524</ref> | '''Acyl carrier protein''' (ACP) is a component of the fatty acid biosynthesis cycle. ACP catalyzes the addition of a thioester to a phosphopantetheine moiety. The phosphopantetheine moiety is added post-translationally to ACP serine residue by ACP synthetase (ACPS). The phosphopantetheine contains a free SH group which binds acyl groups in the fatty acid biosynthesis as thioesters. The acyl groups from acetyl-CoA and malonyl-CoA are transferred to ACP. There are 2 types of ACP. '''ACP I''' is a multifunctional polypeptide found in yeast and mammals while '''ACP II''' is a monomeric protein found in bacteria and plants.<ref>PMID:18059524</ref> | ||
| + | |||
| + | See also: | ||
| + | *[[Molecular Playground/ACP apo]] | ||
| + | *[[Acyl carrier protein synthase]] | ||
| + | *[[Lipid metabolism]] | ||
| + | *[[Fatty acid synthesis]] | ||
== Structural highlights == | == Structural highlights == | ||
| - | <scene name='59/592131/Cv/ | + | <scene name='59/592131/Cv/11'>Biological Assembly of E. coli acyl carrier protein</scene> ([[2fad]]). <scene name='59/592131/Cv/9'>Active site</scene> of ''E. coli'' acyl carrier protein. Water molecules are shown as red spheres. |
| - | The <scene name='59/592131/Cv/ | + | The <scene name='59/592131/Cv/10'>thioester - heptanethioate - is bound to ACP serine residue</scene> (colored in darkmagenta) in an expandable hydrophobic cavity.<ref>PMID:17059829</ref> |
| - | + | *<scene name='59/592131/Cv/15'>Na coordination site</scene>. | |
| - | </ | + | *<scene name='59/592131/Cv/16'>Active site hydrophobic cavity with thioester heptanethioate</scene>. |
==3D structures of acyl carrier protein== | ==3D structures of acyl carrier protein== | ||
| + | [[Acyl carrier protein 3D structures]] | ||
| - | + | </StructureSection> | |
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | + | ||
| - | **[[2fhs]] – EcACP + enoyl-[ACP] reductase <br /> | ||
| - | **[[3ejb]], [[3ejd]], [[3eje]] – EcACP + thioester + p450BioI<br /> | ||
| - | **[[3ny7]] – EcACP + YchM STAS domain <br /> | ||
| - | **[[1f80]] – BsACP + ACPS <br /> | ||
| - | **[[4dxe]] – ACP + ACPS – ''Staphylococcus aureus''<br /> | ||
| - | **[[4etw]] – ACP + pimelyl-[ACP] methyl ester esterase – ''Shigella flexneri''<br /> | ||
| - | **[[4ihf]], [[4ihg]], [[4ihh]] – EcACP + protein FIRA <br /> | ||
| - | **[[4keh]] – EcACP + fatty acid dehydratase <br /> | ||
| - | }} | ||
== References == | == References == | ||
<references/> | <references/> | ||
[[Category:Topic Page]] | [[Category:Topic Page]] | ||
Current revision
| |||||||||||
References
- ↑ Byers DM, Gong H. Acyl carrier protein: structure-function relationships in a conserved multifunctional protein family. Biochem Cell Biol. 2007 Dec;85(6):649-62. PMID:18059524 doi:http://dx.doi.org/10.1139/o07-109
- ↑ Roujeinikova A, Simon WJ, Gilroy J, Rice DW, Rafferty JB, Slabas AR. Structural studies of fatty acyl-(acyl carrier protein) thioesters reveal a hydrophobic binding cavity that can expand to fit longer substrates. J Mol Biol. 2007 Jan 5;365(1):135-45. Epub 2006 Sep 23. PMID:17059829 doi:10.1016/j.jmb.2006.09.049